Antagonism of calcium currents and neurotransmitter release by barium ions at frog motor nerve endings
- PMID: 10694243
- PMCID: PMC1571827
- DOI: 10.1038/sj.bjp.0703036
Antagonism of calcium currents and neurotransmitter release by barium ions at frog motor nerve endings
Abstract
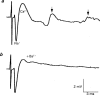
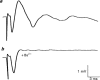
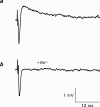
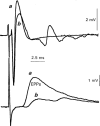
1. The effects of Ba(2+) (0.1 - 2 mM) on the component of the perineural voltage change associated with nerve terminal calcium currents (prejunctional Ca(2+) currents) were compared with the effects of this ion to antagonize calcium-dependent acetylcholine (ACh) release. These experiments were made on isolated neuromuscular junctions of the frog. 2. In the presence of sufficient concentrations of K(+) channel blockers to eliminate measurable prejunctional K(+) currents, low concentrations of Ba(2+) selectively antagonized prejunctional Ca(2+) currents in normal Ca(2+) solutions. Higher concentrations of Ba(2+) also substantially reduced the Na(+) component of the perineural waveform. 3. Ba(2+) inhibited the prolonged prejunctional Ca(2+) currents that developed in the presence of higher concentrations of K(+) channel blockers. 4. Simultaneous measurements of the prejunctional Ca(2+) currents and the electrophysiological correlates of ACh release (i.e. end-plate potentials, EPPs) were made under conditions of modest K(+) channel blockade. Under these conditions, Ba(2+) generally produced simultaneous decreases in both Ca(2+) currents and EPP amplitudes. In some instances, a prolongation of prejunctional Ca(2+) currents and a transient increase in EPP amplitudes preceded the decreases in both electrophysiological events. 5. These results suggest that Ba(2+) ions can antagonize the entry of calcium into motor nerve endings and this effect is likely to be responsible for the inhibitory effects of Ba(2+) on evoked ACh release.
Figures


Similar articles
-
The phosphatidylinositol 4-kinase inhibitor phenylarsine oxide blocks evoked neurotransmitter release by reducing calcium entry through N-type calcium channels.Br J Pharmacol. 2000 May;130(2):418-24. doi: 10.1038/sj.bjp.0703299. Br J Pharmacol. 2000. PMID: 10807681 Free PMC article.
-
Opposing effects of phorbol esters on transmitter release and calcium currents at frog motor nerve endings.J Physiol. 1997 May 15;501 ( Pt 1)(Pt 1):41-8. doi: 10.1111/j.1469-7793.1997.041bo.x. J Physiol. 1997. PMID: 9174992 Free PMC article.
-
Adenosine decreases both presynaptic calcium currents and neurotransmitter release at the mouse neuromuscular junction.J Physiol. 2004 Jul 15;558(Pt 2):389-401. doi: 10.1113/jphysiol.2004.061457. Epub 2004 May 14. J Physiol. 2004. PMID: 15146054 Free PMC article.
-
Influence of heavy metals on synaptic transmission: a review.Neurotoxicology. 1983 Winter;4(4):69-83. Neurotoxicology. 1983. PMID: 6322059 Review.
-
Prejunctional actions of muscle relaxants: synaptic vesicles and transmitter mobilization as sites of action.Gen Pharmacol. 1995 Jul;26(4):659-66. doi: 10.1016/0306-3623(94)00246-j. Gen Pharmacol. 1995. PMID: 7635241 Review.
Cited by
-
The phosphatidylinositol 4-kinase inhibitor phenylarsine oxide blocks evoked neurotransmitter release by reducing calcium entry through N-type calcium channels.Br J Pharmacol. 2000 May;130(2):418-24. doi: 10.1038/sj.bjp.0703299. Br J Pharmacol. 2000. PMID: 10807681 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

