A phosphotyrosine displacement mechanism for activation of Src by PTPalpha
- PMID: 10698938
- PMCID: PMC305636
- DOI: 10.1093/emboj/19.5.964
A phosphotyrosine displacement mechanism for activation of Src by PTPalpha
Abstract
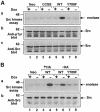
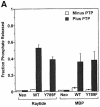
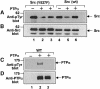
Protein tyrosine phosphatase alpha (PTPalpha) is believed to dephosphorylate physiologically the Src proto-oncogene at phosphotyrosine (pTyr)527, a critical negative-regulatory residue. It thereby activates Src, and PTPalpha overexpression neoplastically transforms NIH 3T3 cells. pTyr789 in PTPalpha is constitutively phosphorylated and binds Grb2, an interaction that may inhibit PTPalpha activity. We show here that this phosphorylation also specifically enables PTPalpha to dephosphorylate pTyr527. Tyr789-->Phe mutation abrogates PTPalpha-Src binding, dephosphorylation of pTyr527 (although not of other substrates), and neoplastic transformation by overexpressed PTPalpha in vivo. We suggest that pTyr789 enables pTyr527 dephosphorylation by a pilot binding with the Src SH2 domain that displaces the intramolecular pTyr527-SH2 binding. Consistent with model predictions, we find that excess SH2 domains can disrupt PTPalpha-Src binding and can block PTPalpha-mediated dephosphorylation and activation in proportion to their affinity for pTyr789. Moreover, we show that, as predicted by the model, catalytically defective PTPalpha has reduced Src binding in vivo. The displacement mechanism provides another potential control point for physiological regulation of Src-family signal transduction pathways.
Figures












References
-
- Alema S., Cassalbore, P., Agostini, E. and Tato, F. (1985) Differentiation of PC12 phaeochromocytoma cells induced by v-src oncogene. Nature, 316, 557–559. - PubMed
-
- Bagrodia S., Chackalaparampil, I., Kmiecik, T.E. and Shalloway, D. (1991) Altered tyrosine 527 phosphorylation and mitotic activation of p60c-src. Nature, 349, 172–175. - PubMed
-
- Bagrodia S., Laudano, A.P. and Shalloway, D. (1994) Accessibility of the c-src SH2-domain for binding is increased during mitosis. J. Biol. Chem., 269, 10247–10251. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

