Chimeric dengue type 2 (vaccine strain PDK-53)/dengue type 1 virus as a potential candidate dengue type 1 virus vaccine
- PMID: 10708416
- PMCID: PMC111800
- DOI: 10.1128/jvi.74.7.3020-3028.2000
Chimeric dengue type 2 (vaccine strain PDK-53)/dengue type 1 virus as a potential candidate dengue type 1 virus vaccine
Abstract
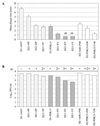
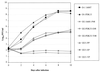
We constructed chimeric dengue type 2/type 1 (DEN-2/DEN-1) viruses containing the nonstructural genes of DEN-2 16681 virus or its vaccine derivative, strain PDK-53, and the structural genes (encoding capsid protein, premembrane protein, and envelope glycoprotein) of DEN-1 16007 virus or its vaccine derivative, strain PDK-13. We previously reported that attenuation markers of DEN-2 PDK-53 virus were encoded by genetic loci located outside the structural gene region of the PDK-53 virus genome. Chimeric viruses containing the nonstructural genes of DEN-2 PDK-53 virus and the structural genes of the parental DEN-1 16007 virus retained the attenuation markers of small plaque size and temperature sensitivity in LLC-MK(2) cells, less efficient replication in C6/36 cells, and attenuation for mice. These chimeric viruses elicited higher mouse neutralizing antibody titers against DEN-1 virus than did the candidate DEN-1 PDK-13 vaccine virus or chimeric DEN-2/DEN-1 viruses containing the structural genes of the PDK-13 virus. Mutations in the envelope protein of DEN-1 PDK-13 virus affected in vitro phenotype and immunogenicity in mice. The current PDK-13 vaccine is the least efficient of the four Mahidol candidate DEN virus vaccines in human trials. The chimeric DEN-2/DEN-1 virus might be a potential DEN-1 virus vaccine candidate. This study indicated that the infectious clones derived from the candidate DEN-2 PDK-53 vaccine are promising attenuated vectors for development of chimeric flavivirus vaccines.
Figures

References
-
- Bancroft W H, Scott R M, Brandt W E, McCown J M, Eckels K H, Hayes D E, Gould D J, Russell P K. Dengue-2 vaccine: infection of Aedes aegypti mosquitoes by feeding on viremic recipients. Am J Trop Med Hyg. 1982;31:1229–1231. - PubMed
-
- Barrett A D T, Mathews J H, Miller B R, Medlen A R, Ledger T N, Roehrig J T. Identification of monoclonal antibodies that distinguish between 17D-204 and other strains of yellow fever virus. J Gen Virol. 1990;71:13–18. - PubMed
-
- Bhamarapravati N, Yoksan S. Live attenuated tetravalent vaccine. In: Gubler D J, Kuno G, editors. Dengue and dengue hemorrhagic fever. Wallingford, United Kingdom: CAB International; 1997. pp. 367–377.
-
- Bhamarapravati N, Yoksan S. Study of bivalent dengue vaccine in volunteers. Lancet. 1989;i:1077. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

