Putative fusogenic activity of NSF is restricted to a lipid mixture whose coalescence is also triggered by other factors
- PMID: 10716927
- PMCID: PMC305668
- DOI: 10.1093/emboj/19.6.1272
Putative fusogenic activity of NSF is restricted to a lipid mixture whose coalescence is also triggered by other factors
Abstract
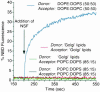
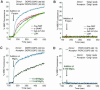
It has recently been reported that N-ethylmaleimide-sensitive fusion ATPase (NSF) can fuse protein-free liposomes containing substantial amounts of 1,2-dioleoylphosphatidylserine (DOPS) and 1, 2-dioleoyl-phosphatidyl-ethanolamine (DOPE) (Otter-Nilsson et al., 1999). The authors impart physiological significance to this observation and propose to re-conceptualize the general role of NSF in fusion processes. We can confirm that isolated NSF can fuse liposomes of the specified composition. However, this activity of NSF is resistant to inactivation by N-ethylmaleimide and does not depend on the presence of alpha-SNAP (soluble NSF-attachment protein). Moreover, under the same conditions, either alpha-SNAP, other proteins apparently unrelated to vesicular transport (glyceraldehyde-3-phosphate dehydrogenase or lactic dehydrogenase) or even 3 mM magnesium ions can also cause lipid mixing. In contrast, neither NSF nor the other proteins nor magnesium had any significant fusogenic activity with liposomes composed of a biologically occurring mixture of lipids. A straightforward explanation is that the lipid composition chosen as optimal for NSF favors non-specific fusion because it is physically unstable when formed into liposomes. A variety of minor perturbations could then trigger coalescence.
Figures


References
-
- Balch W.E., Dunphy, W.G., Braell, W.A. and Rothman, J.E. (1984) Reconstitution of the transport of protein between successive compartments of the Golgi measured by the coupled incorporation of N-acetylglucosamine. Cell, 39, 405–416. - PubMed
-
- Banerjee A., Barry,V.A., DasGupta,B.R. and Martin,T.F.J. (1996) N–ethylmaleimide-sensitive factor acts at a prefusion ATP-dependent step in Ca2+-activated exocytosis. J. Biol. Chem., 271, 20223–20226. - PubMed
-
- Beckers C.J., Block, M.R., Glick, B.S., Rothman, J.E. and Balch, W.E. (1989) Vesicular transport between the endoplasmic reticulum and the Golgi stack requires the NEM-sensitive fusion protein. Nature, 339, 397–398. - PubMed
-
- Colombo M.I., Taddese, M., Whiteheart, S.W. and Stahl, P.D. (1996) A possible predocking attachment site for N–ethylmaleimide-sensitive fusion protein. Insights from in vitro endosome fusion. J. Biol. Chem., 271, 18810–18816. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

