Influence of bilirubin and other antioxidants on nitrergic relaxation in the pig gastric fundus
- PMID: 10725269
- PMCID: PMC1571952
- DOI: 10.1038/sj.bjp.0703176
Influence of bilirubin and other antioxidants on nitrergic relaxation in the pig gastric fundus
Abstract
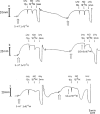
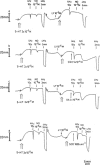
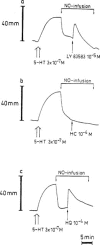
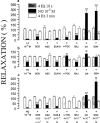
1. The influence of several antioxidants (bilirubin, urate, ascorbate, alpha-tocopherol, glutathione (GSH), Cu/Zn superoxide dismutase (SOD) and the manganese SOD mimic EUK-8) on nitrergic relaxations induced by either exogenous nitric oxide (NO; 10(-5) M) or electrical field stimulation (4 Hz; 10 s and 3 min) was studied in the pig gastric fundus. 2. Ascorbate (5x10(-4) M), alpha-tocopherol (4x10(-4) M), SOD (300 - 1000 u ml(-1)) and EUK-8 (3x10(-4) M) did not influence the relaxations to exogenous NO. In the presence of GSH (5x10(-4) M), the short-lasting relaxation to NO became biphasic, potentiated and prolonged. Urate (4x10(-4) M) and bilirubin (2x10(-4) M) also potentiated the relaxant effect of NO. None of the antioxidants influenced the electrically evoked relaxations. 3. 6-Anilino-5,8-quinolinedione (LY83583; 10(-5) M) had no influence on nitrergic nerve stimulation but nearly abolished the relaxant response to exogenous NO. Urate and GSH completely prevented this inhibitory effect, while it was partially reversed by SOD and bilirubin. Ascorbate, alpha-tocopherol and EUK-8 were without effect. 4. Hydroquinone (10(-4) M) did not affect the electrically induced nitrergic relaxations, but markedly reduced NO-induced relaxations. The inhibition of exogenous NO by hydroquinone was completely prevented by urate and GSH. SOD and ascorbate afforded partial protection, while bilirubin, EUK-8 and alpha-tocopherol were ineffective. 5. Hydroxocobalamin (10(-4) M) inhibited relaxations to NO by 50%, but not the electrically induced responses. Full protection versus this inhibitory effect was obtained with urate, GSH and alpha-tocopherol. 6. These results strengthen the hypothesis that several endogenous antioxidant defense mechanisms, enzymatic as well as non-enzymatic, might play a role in the nitrergic neurotransmission process.
Figures



Similar articles
-
Influence of antioxidant depletion on nitrergic relaxation in the pig gastric fundus.Br J Pharmacol. 2002 Feb;135(4):917-26. doi: 10.1038/sj.bjp.0704553. Br J Pharmacol. 2002. PMID: 11861319 Free PMC article.
-
Interaction of hypoxanthine/xanthine oxidase with nitrergic relaxation in the porcine gastric fundus.Br J Pharmacol. 2000 May;130(2):359-66. doi: 10.1038/sj.bjp.0703317. Br J Pharmacol. 2000. PMID: 10807674 Free PMC article.
-
Antioxidant protection of NO-induced relaxations of the mouse anococcygeus against inhibition by superoxide anions, hydroquinone and carboxy-PTIO.Br J Pharmacol. 1996 Sep;119(2):432-8. doi: 10.1111/j.1476-5381.1996.tb16004.x. Br J Pharmacol. 1996. PMID: 8886431 Free PMC article.
-
Role of antioxidants in the protection of the nitrergic neurotransmitter.Acta Neurol Belg. 2002 Jun;102(2):68-72. Acta Neurol Belg. 2002. PMID: 12161902 Review.
-
Superoxide anions, free-radical scavengers, and nitrergic neurotransmission.Gen Pharmacol. 1997 Apr;28(4):489-93. doi: 10.1016/s0306-3623(96)00281-9. Gen Pharmacol. 1997. PMID: 9147013 Review.
Cited by
-
Pre- and postjunctional protective effect of neocuproine on the nitrergic neurotransmitter in the mouse gastric fundus.Br J Pharmacol. 2001 Jan;132(1):277-85. doi: 10.1038/sj.bjp.0703772. Br J Pharmacol. 2001. PMID: 11156587 Free PMC article.
-
Influence of antioxidant depletion on nitrergic relaxation in the pig gastric fundus.Br J Pharmacol. 2002 Feb;135(4):917-26. doi: 10.1038/sj.bjp.0704553. Br J Pharmacol. 2002. PMID: 11861319 Free PMC article.
-
The role of carbon monoxide in the gastrointestinal tract.J Physiol. 2004 Apr 15;556(Pt 2):325-36. doi: 10.1113/jphysiol.2003.056556. Epub 2004 Feb 6. J Physiol. 2004. PMID: 14766943 Free PMC article. Review.
-
In vitro analysis of the effect of hyperbilirubinemia on rabbit ureter and bladder.Pediatr Nephrol. 2006 Mar;21(3):328-32. doi: 10.1007/s00467-005-2094-3. Epub 2005 Dec 29. Pediatr Nephrol. 2006. PMID: 16382325
-
Interaction of hypoxanthine/xanthine oxidase with nitrergic relaxation in the porcine gastric fundus.Br J Pharmacol. 2000 May;130(2):359-66. doi: 10.1038/sj.bjp.0703317. Br J Pharmacol. 2000. PMID: 10807674 Free PMC article.
References
-
- ABRAMOVITZ M., HOMMA H., ISHIGAKI S., TANSEY F., CAMMER W., LISTOWSKY I. Characterization and localization of glutathione-S-transferases in rat brain and binding of hormones, neurotransmitters, and drugs. J. Neurochem. 1988;50:50–57. - PubMed
-
- BARBIER A.J.M., LEFEBVRE R.A. Effect of LY83583 on relaxation induced by non-adrenergic non-cholinergic nerve stimulation and exogenous nitric oxide in the rat gastric fundus. Eur. J. Pharmacol. 1992;219:331–334. - PubMed
-
- BECKER B.F. Towards the physiological function of uric acid. Free Rad. Biol. Med. 1993;14:615–631. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

