Cystic fibrosis transmembrane conductance regulator does not affect neutrophil migration across cystic fibrosis airway epithelial monolayers
- PMID: 10751364
- PMCID: PMC1876904
- DOI: 10.1016/S0002-9440(10)65009-2
Cystic fibrosis transmembrane conductance regulator does not affect neutrophil migration across cystic fibrosis airway epithelial monolayers
Erratum in
- Am J Pathol 2000 Oct;157(4):1413
Abstract
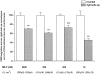
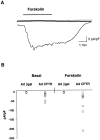
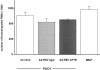
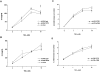
Recent studies have shown that airway inflammation dominated by neutrophils, ie, polymorphonuclear cells (PMN) was observed in infants and children with cystic fibrosis (CF) even in the absence of detectable infection. To assess whether there is a CF-related anomaly of PMN migration across airway epithelial cells, we developed an in vitro model of chemotactic migration across tight and polarized CF(15) cells, a CF human nasal epithelial cell line, seeded on porous filters. To compare PMN migration across a pair of CF and control monolayers in the physiological direction, inverted CF(15) cells were infected with increasing concentrations of recombinant adenoviruses containing either the normal cystic fibrosis transmembrane conductance regulator (CFTR) cDNA, the DeltaF508 CFTR cDNA, or the beta-galactosidase gene. The number of PMN migrating in response to N-formyl-Met-Leu-Phe across inverted CF(15) monolayers expressing beta-galactosidase was similar to that seen across CF(15) monolayers rescued with CFTR, whatever the proportion of cells expressing the transgene. Moreover, PMN migration across monolayers expressing various amounts of mutated CFTR was not different from that observed across matched counterparts expressing normal CFTR. Finally, PMN migration in response to adherent or Pseudomonas aeruginosa was equivalent across CF and corrected monolayers. The possibility that mutated CFTR may exert indirect effects on PMN recruitment, via an abnormal production of the chemotactic cytokine interleukin-8, was also explored. Apical and basolateral production of interleukin-8 by polarized CF cells expressing mutated CFTR was not different from that observed with rescued cells, either in baseline or stimulated conditions. CF(15) cells displayed a CF phenotype that could be corrected by CFTR-containing adenoviruses, because two known CF defects, Cl(-) secretion and increased P. aeruginosa adherence, were normalized after infection with those viruses. Thus, we conclude that the presence of a mutated CFTR does not per se lead to an exaggerated inflammatory response of CF surface epithelial cells in the absence or presence of a bacterial infection.
Figures




References
-
- Suter S, Schaad UB, Tegner H, Ohlsson K, Desgrandchamps D, Waldvogel F: Free granulocyte elastase levels in bronchial secretions from patients with cystic fibrosis: effect of antimicrobial treatment against Pseudomonas aeruginosa. J Infect Dis 1986, 153:902-907 - PubMed
-
- Goldstein W, Döring G: Lysosomal enzymes from polymorphonuclear leukocytes and proteinase inhibitors in patients with cystic fibrosis. Am Rev Respir Dis 1986, 134:49-56 - PubMed
-
- Pilewski JM, Frizzell RA: Role of CFTR in airway disease. Physiol Rev 1999, 79:S215-S255 - PubMed
-
- Balough K, McCubbin M, Weinberger M, Smits W, Ahrens R, Fick R: The relationship between infection and inflammation in the early stages of lung disease from cystic fibrosis. Pediatr Pulmonol 1995, 20:63-70 - PubMed
-
- Khan TZ, Wagener JS, Bost T, Martinez J, Accurso FJ, Riches DWH: Early pulmonary inflammation in infants with cystic fibrosis. Am J Respir Crit Care Med 1995, 151:1075-1082 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

