Bean alpha-amylase inhibitor 1 in transgenic peas (Pisum sativum) provides complete protection from pea weevil (Bruchus pisorum) under field conditions
- PMID: 10759552
- PMCID: PMC18100
- DOI: 10.1073/pnas.070054597
Bean alpha-amylase inhibitor 1 in transgenic peas (Pisum sativum) provides complete protection from pea weevil (Bruchus pisorum) under field conditions
Abstract
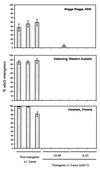
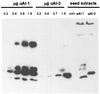
Two alpha-amylase inhibitors, called alphaAI-1 and alphaAI-2, that share 78% amino acid sequence identity and have a differential specificity toward mammalian and insect alpha-amylases are present in different accessions of the common bean (Phaseolus vulgaris). Using greenhouse-grown transgenic peas (Pisum sativum), we have shown previously that expression of alphaAI-1 in pea seeds can provide complete protection against the pea weevil (Bruchus pisorum). Here, we report that alphaAI-1 also protects peas from the weevil under field conditions. The high degree of protection is explained by our finding that alphaAI-1 inhibits pea bruchid alpha-amylase by 80% over a broad pH range (pH 4.5-6.5). alphaAI-2, on the other hand, is a much less effective inhibitor of pea bruchid alpha-amylase, inhibiting the enzyme by only 40%, and only in the pH 4.0-4.5 range. Nevertheless, this inhibitor was still partially effective in protecting field-grown transgenic peas against pea weevils. The primary effect of alphaAI-2 appeared to be a delay in the maturation of the larvae. This contrasts with the effect of alphaAI-1, which results in larval mortality at the first or second instar. These results are discussed in relationship to the use of amylase inhibitors with different specificities to bring about protection of crops from their insect pests or to decrease insect pest populations below the economic injury level.
Figures



References
-
- Roush R T, Shelton A M. Nat Biotech. 1997;15:816–817. - PubMed
-
- Reek G R, Kramer K J, Baker J E, Kanost M R, Fabrick J A, Behnke C A. In: Advances in Insect Control: The Role of Transgenic Plants. Carozzi N, Koziel M, editors. London: Taylor & Francis; 1997. pp. 157–183.
-
- Chrispeels M J. In: Advances in Insect Control. The Role of Transgenic Plants. Carozzi N, Koziel M, editors. London: Taylor & Francis; 1997. pp. 139–156.
-
- Ho M F, Whitaker J R. J Food Biochem. 1993;17:15–33.
-
- Ho M F, Whitaker J R. J Food Biochem. 1993;17:35–52.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

