Conversion of alpha-lactalbumin to a protein inducing apoptosis
- PMID: 10760289
- PMCID: PMC18203
- DOI: 10.1073/pnas.97.8.4221
Conversion of alpha-lactalbumin to a protein inducing apoptosis
Abstract
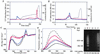
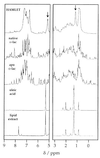
In this study alpha-lactalbumin was converted from the regular, native state to a folding variant with altered biological function. The folding variant was shown to induce apoptosis in tumor cells and immature cells, but healthy cells were resistant to this effect. Conversion to HAMLET (human alpha-lactalbumin made lethal to tumor cells) required partial unfolding of the protein and a specific fatty acid, C18:1, as a necessary cofactor. Conversion was achieved with alpha-lactalbumin derived from human milk whey and with recombinant protein expressed in Escherichia coli. We thus have identified the folding change and the fatty acid as two key elements that define HAMLET, the apoptosis-inducing functional state of alpha-lactalbumin. Although the environment in the mammary gland favors the native conformation of alpha-lactalbumin that serves as a specifier in the lactose synthase complex, the conditions under which HAMLET was formed resemble those in the stomach of the nursing child. Low pH is known to release Ca(2+) from the high-affinity Ca(2+)-binding site and to activate lipases that hydrolyze free fatty acids from milk triglycerides. We propose that this single amino acid polypeptide chain may perform vastly different biological functions depending on its folding state and the in vivo environment. It may be speculated that molecules like HAMLET can aid in lowering the incidence of cancer in breast-fed children by purging of tumor cells from the gut of the neonate.
Figures


References
-
- Booth D R, Sunde M, Bellotti V, Robinson C V, Hutchinson W L, Fraser P E, Hawkins P N, Dobson C M, Radford S E, Blake C C, et al. Nature (London) 1997;385:787–793. - PubMed
-
- Herrmann J M, Malkus P, Schekman R. Trends Cell Biol. 1999;9:5–7. - PubMed
-
- Martin J, Hartl F U. Curr Opin Struct Biol. 1997;7:41–52. - PubMed
-
- Safar J, Roller P, Gajdusek D, Gibbs C J. J Biol Chem. 1993;268:20276–20284. - PubMed
-
- Safar J, Roller P, Gajdusek D, Gibbs C J. Biochemistry. 1994;33:8375–8383. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

