A rac homolog is required for induction of hyphal growth in the dimorphic yeast Yarrowia lipolytica
- PMID: 10762235
- PMCID: PMC111297
- DOI: 10.1128/JB.182.9.2376-2386.2000
A rac homolog is required for induction of hyphal growth in the dimorphic yeast Yarrowia lipolytica
Abstract
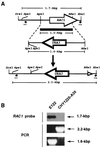
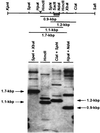
Dimorphism in fungi is believed to constitute a mechanism of response to adverse conditions and represents an important attribute for the development of virulence by a number of pathogenic fungal species. We have isolated YlRAC1, a gene encoding a 192-amino-acid protein that is essential for hyphal growth in the dimorphic yeast Yarrowia lipolytica and which represents the first Rac homolog described for fungi. YlRAC1 is not an essential gene, and its deletion does not affect the ability to mate or impair actin polarization in Y. lipolytica. However, strains lacking functional YlRAC1 show alterations in cell morphology, suggesting that the function of YlRAC1 may be related to some aspect of the polarization of cell growth. Northern blot analysis showed that transcription of YlRAC1 increases steadily during the yeast-to-hypha transition, while Southern blot analysis of genomic DNA suggested the presence of several RAC family members in Y. lipolytica. Interestingly, strains lacking functional YlRAC1 are still able to grow as the pseudohyphal form and to invade agar, thus pointing to a function for YlRAC1 downstream of MHY1, a previously isolated gene encoding a C(2)H(2)-type zinc finger protein with the ability to bind putative stress response elements and whose activity is essential for both hyphal and pseudohyphal growth in Y. lipolytica.
Figures









Similar articles
-
Isolation and characterization of YlBEM1, a gene required for cell polarization and differentiation in the dimorphic yeast Yarrowia lipolytica.Eukaryot Cell. 2002 Aug;1(4):526-37. doi: 10.1128/EC.1.4.526-537.2002. Eukaryot Cell. 2002. PMID: 12456001 Free PMC article.
-
YlBMH1 encodes a 14-3-3 protein that promotes filamentous growth in the dimorphic yeast Yarrowia lipolytica.Microbiology (Reading). 2002 Nov;148(Pt 11):3725-3735. doi: 10.1099/00221287-148-11-3725. Microbiology (Reading). 2002. PMID: 12427962
-
MHY1 encodes a C2H2-type zinc finger protein that promotes dimorphic transition in the yeast Yarrowia lipolytica.J Bacteriol. 1999 May;181(10):3051-7. doi: 10.1128/JB.181.10.3051-3057.1999. J Bacteriol. 1999. PMID: 10322005 Free PMC article.
-
HOY1, a homeo gene required for hyphal formation in Yarrowia lipolytica.Mol Cell Biol. 1997 Nov;17(11):6283-93. doi: 10.1128/MCB.17.11.6283. Mol Cell Biol. 1997. PMID: 9343389 Free PMC article.
-
Physiology and genetics of the dimorphic fungus Yarrowia lipolytica.FEMS Microbiol Rev. 1997 Apr;19(4):219-37. doi: 10.1111/j.1574-6976.1997.tb00299.x. FEMS Microbiol Rev. 1997. PMID: 9167256 Review.
Cited by
-
Rac1 and Cdc42 have different roles in Candida albicans development.Eukaryot Cell. 2006 Feb;5(2):321-9. doi: 10.1128/EC.5.2.321-329.2006. Eukaryot Cell. 2006. PMID: 16467473 Free PMC article.
-
Copper Ion Mediates Yeast-to-Hypha Transition in Yarrowia lipolytica.J Fungi (Basel). 2023 Feb 13;9(2):249. doi: 10.3390/jof9020249. J Fungi (Basel). 2023. PMID: 36836363 Free PMC article.
-
A Rac homolog functions downstream of Ras1 to control hyphal differentiation and high-temperature growth in the pathogenic fungus Cryptococcus neoformans.Eukaryot Cell. 2005 Jun;4(6):1066-78. doi: 10.1128/EC.4.6.1066-1078.2005. Eukaryot Cell. 2005. PMID: 15947199 Free PMC article.
-
GTPase Rac Regulates Conidiation, AFB1 Production and Stress Response in Pathogenic Fungus Aspergillus flavus.Toxins (Basel). 2022 Aug 24;14(9):581. doi: 10.3390/toxins14090581. Toxins (Basel). 2022. PMID: 36136519 Free PMC article.
-
Yarrowia lipolytica: a beneficious yeast in biotechnology as a rare opportunistic fungal pathogen: a minireview.World J Microbiol Biotechnol. 2018 Dec 21;35(1):10. doi: 10.1007/s11274-018-2583-8. World J Microbiol Biotechnol. 2018. PMID: 30578432 Free PMC article. Review.
References
-
- Adams A E M, Pringle J R. Staining of actin with fluorochrome-conjugated phalloidin. Methods Enzymol. 1991;194:729–731. - PubMed
-
- Akashi T, Kanbe T, Tanaka K. The role of the cytoskeleton in the polarized growth of the germ tube in in Candida albicans. Microbiology. 1994;140:271–280. - PubMed
-
- Alfa C E, Hyams J S. Distribution of tubulin and actin through the cell division cycle of the fission yeast Schizosaccharomyces japonicus var. versitalis: comparison with Schizosaccharomyces pombe. J Cell Sci. 1990;96:71–77. - PubMed
-
- Arellano M, Coll P M, Perez P. RHO GTPases in the control of cell morphology, cell polarity, and actin localization in fission yeast. Microsc Res Tech. 1999;47:51–60. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

