Humanized in vivo model for streptococcal impetigo
- PMID: 10768985
- PMCID: PMC97500
- DOI: 10.1128/IAI.68.5.2880-2887.2000
Humanized in vivo model for streptococcal impetigo
Abstract
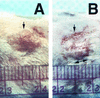
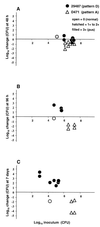
An in vivo model for group A streptococcal (GAS) impetigo was developed, whereby human neonatal foreskin engrafted onto SCID mice was superficially damaged and bacteria were topically applied. Severe infection, indicated by a purulent exudate, could be induced with as few as 1,000 CFU of a virulent strain. Early findings (48 h) showed a loss of stratum corneum and adherence of short chains of gram-positive cocci to the external surface of granular keratinocytes. This was followed by an increasing infiltration of polymorphonuclear leukocytes (neutrophils) of mouse origin, until a thick layer of pus covered an intact epidermis, with massive clumps of cocci accumulated at the outer rim of the pus layer. By 7 days postinoculation, the epidermis was heavily eroded; in some instances, the dermis contained pockets (ulcers) filled with cocci, similar to that observed for ecthyma. Importantly, virulent GAS underwent reproduction, resulting in a net increase in CFU of 20- to 14,000-fold. The majority of emm pattern D strains had a higher gross pathology score than emm pattern A, B, or C (A-C) strains, consistent with epidemiological findings that pattern D strains have a strong tendency to cause impetigo, whereas pattern A-C strains are more likely to cause pharyngitis.
Figures



References
-
- Bessen D, Fischetti V A. A human IgG receptor of group A streptococci is associated with tissue site of infection and streptococcal class. J Infect Dis. 1990;161:747–754. - PubMed
-
- Bessen D, Fischetti V A. Synthetic peptide vaccine against mucosal colonization by group A streptococci. I. Protection against a heterologous M serotype with shared C repeat region epitopes. J Immunol. 1990;145:1251–1256. - PubMed
-
- Bessen D E, Izzo M W, Fiorentino T R, Caringal R M, Hollingshead S K, Beall B. Genetic linkage of exotoxin alleles and emm gene markers for tissue tropism in group A streptococci. J Infect Dis. 1999;179:627–636. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

