Keratins turn over by ubiquitination in a phosphorylation-modulated fashion
- PMID: 10791969
- PMCID: PMC2174842
- DOI: 10.1083/jcb.149.3.547
Keratins turn over by ubiquitination in a phosphorylation-modulated fashion
Abstract
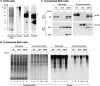
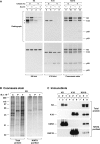
Keratin polypeptides 8 and 18 (K8/18) are intermediate filament (IF) proteins that are expressed in glandular epithelia. Although the mechanism of keratin turnover is poorly understood, caspase-mediated degradation of type I keratins occurs during apoptosis and the proteasome pathway has been indirectly implicated in keratin turnover based on colocalization of keratin-ubiquitin antibody staining. Here we show that K8 and K18 are ubiquitinated based on cotransfection of His-tagged ubiquitin and human K8 and/or K18 cDNAs, followed by purification of ubiquitinated proteins and immunoblotting with keratin antibodies. Transfection of K8 or K18 alone yields higher levels of keratin ubiquitination as compared with cotransfection of K8/18, likely due to stabilization of the keratin heteropolymer. Most of the ubiquitinated species partition with the noncytosolic keratin fraction. Proteasome inhibition stabilizes K8 and K18 turnover, and is associated with accumulation of phosphorylated keratins, which indicates that although keratins are stable they still turnover. Analysis of K8 and K18 ubiquitination and degradation showed that K8 phosphorylation contributes to its stabilization. Our results provide direct evidence for K8 and K18 ubiquitination, in a phosphorylation modulated fashion, as a mechanism for regulating their turnover and suggest that other IF proteins could undergo similar regulation. These and other data offer a model that links keratin ubiquitination and hyperphosphorylation that, in turn, are associated with Mallory body deposits in a variety of liver diseases.
Figures


Similar articles
-
Keratin hypersumoylation alters filament dynamics and is a marker for human liver disease and keratin mutation.J Biol Chem. 2011 Jan 21;286(3):2273-84. doi: 10.1074/jbc.M110.171314. Epub 2010 Nov 9. J Biol Chem. 2011. PMID: 21062750 Free PMC article.
-
Dynamics of human keratin 18 phosphorylation: polarized distribution of phosphorylated keratins in simple epithelial tissues.J Cell Biol. 1995 Dec;131(5):1291-301. doi: 10.1083/jcb.131.5.1291. J Cell Biol. 1995. PMID: 8522590 Free PMC article.
-
Protein phosphatase inhibition in normal and keratin 8/18 assembly-incompetent mouse strains supports a functional role of keratin intermediate filaments in preserving hepatocyte integrity.Hepatology. 1998 Jul;28(1):116-28. doi: 10.1002/hep.510280117. Hepatology. 1998. PMID: 9657104
-
Keratin modifications and solubility properties in epithelial cells and in vitro.Subcell Biochem. 1998;31:105-40. Subcell Biochem. 1998. PMID: 9932491 Review.
-
Keratins let liver live: Mutations predispose to liver disease and crosslinking generates Mallory-Denk bodies.Hepatology. 2007 Nov;46(5):1639-49. doi: 10.1002/hep.21976. Hepatology. 2007. PMID: 17969036 Review.
Cited by
-
Intermediate filaments control the intracellular distribution of caspases during apoptosis.Am J Pathol. 2004 Feb;164(2):395-407. doi: 10.1016/S0002-9440(10)63130-6. Am J Pathol. 2004. PMID: 14742246 Free PMC article.
-
Characterization of in vivo keratin 19 phosphorylation on tyrosine-391.PLoS One. 2010 Oct 25;5(10):e13538. doi: 10.1371/journal.pone.0013538. PLoS One. 2010. PMID: 21049038 Free PMC article.
-
Autophagy and KRT8/keratin 8 protect degeneration of retinal pigment epithelium under oxidative stress.Autophagy. 2017 Feb;13(2):248-263. doi: 10.1080/15548627.2016.1256932. Epub 2017 Jan 3. Autophagy. 2017. PMID: 28045574 Free PMC article.
-
Revisiting the significance of keratin expression in complex epithelia.J Cell Sci. 2022 Oct 15;135(20):jcs260594. doi: 10.1242/jcs.260594. Epub 2022 Oct 26. J Cell Sci. 2022. PMID: 36285538 Free PMC article.
-
Ubiquitin-immunoreactive degradation products of cytokeratin 8/18 correlate with aggressive breast cancer.Cancer Sci. 2003 Oct;94(10):864-70. doi: 10.1111/j.1349-7006.2003.tb01368.x. Cancer Sci. 2003. PMID: 14556659 Free PMC article.
References
-
- Akamatsu M., Hori S., Tsutsumi Y., Osamura R.Y., Ohkido M. Ubiquitinated cytokeratin inclusions in lichen amyloidosusan immunohistochemical analysis. Pathol. Int. 1995;45:116–122. - PubMed
-
- Cadrin M., Anderson N.M., Aasheim L.H., Kawahara H., Franks D.J., French S.W. Modifications in cytokeratin and actin in cultured liver cells derived from griseofulvin-fed mice. Lab. Invest. 1995;72:453–460. - PubMed
-
- Chipev C.C., Korge B.P., Markova N., Bale S.J., DiGiovanna J.J., Compton J.G., Steinert P.M. A leucine→proline mutation in the H1 subdomain of keratin 1 causes epidermolytic hyperkeratosis. Cell. 1992;70:821–828. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

