Isoforms of alpha-actinin from cardiac, smooth, and skeletal muscle form polar arrays of actin filaments
- PMID: 10791977
- PMCID: PMC2174853
- DOI: 10.1083/jcb.149.3.635
Isoforms of alpha-actinin from cardiac, smooth, and skeletal muscle form polar arrays of actin filaments
Abstract
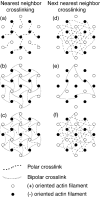
We have used a positively charged lipid monolayer to form two-dimensional bundles of F-actin cross-linked by alpha-actinin to investigate the relative orientation of the actin filaments within them. This method prevents growth of the bundles perpendicular to the monolayer plane, thereby facilitating interpretation of the electron micrographs. Using alpha-actinin isoforms isolated from the three types of vertebrate muscle, i.e., cardiac, skeletal, and smooth, we have observed almost exclusively cross-linking between polar arrays of filaments, i.e., actin filaments with their plus ends oriented in the same direction. One type of bundle can be classified as an Archimedian spiral consisting of a single actin filament that spirals inward as the filament grows and the bundle is formed. These spirals have a consistent hand and grow to a limiting internal diameter of 0.4-0.7 microm, where the filaments appear to break and spiral formation ceases. These results, using isoforms usually characterized as cross-linkers of bipolar actin filament bundles, suggest that alpha-actinin is capable of cross-linking actin filaments in any orientation. Formation of specifically bipolar or polar filament arrays cross-linked by alpha-actinin may require additional factors that either determine the filament orientation or restrict the cross-linking capabilities of alpha-actinin.
Figures





References
-
- Arai Y., Yasuda R., Akashi K., Harada Y., Miyata H., Kinosita K., Itoh H. Tying a molecular knot with optical tweezers. Nature. 1999;399:446–448. - PubMed
-
- Atsuta F., Sato K., Maruyama K., Shimada Y. Distribution of connectin (titin), nebulin and α-actinin at myotendinous junctions of chicken pectoralis musclesan immunofluorescence and immunoelectron microscopic study. J. Muscle Res. Cell Motil. 1993;14:511–517. - PubMed
-
- Blanchard A., Ohanian V., Critchley D. The structure and function of α-actinin. J. Muscle Res. Cell Motil. 1989;10:280–289. - PubMed
-
- Burn P., Rotman A., Meyer R.K., Burger M.M. Diacylglycerol in large α-actinin/actin complexes and the cytoskeleton of activated platelets. Nature. 1985;314:469–472. - PubMed

