Drosophila beta spectrin functions independently of alpha spectrin to polarize the Na,K ATPase in epithelial cells
- PMID: 10791978
- PMCID: PMC2174857
- DOI: 10.1083/jcb.149.3.647
Drosophila beta spectrin functions independently of alpha spectrin to polarize the Na,K ATPase in epithelial cells
Abstract
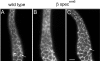
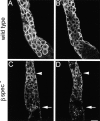
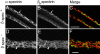
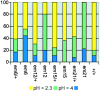
Spectrin has been proposed to function as a sorting machine that concentrates interacting proteins such as the Na,K ATPase within specialized plasma membrane domains of polarized cells. However, little direct evidence to support this model has been obtained. Here we used a genetic approach to directly test the requirement for the beta subunit of the alphabeta spectrin molecule in morphogenesis and function of epithelial cells in Drosophila. beta Spectrin mutations were lethal during late embryonic/early larval development and they produced subtle defects in midgut morphology and stomach acid secretion. The polarized distributions of alphabeta(H) spectrin and ankyrin were not significantly altered in beta spectrin mutants, indicating that the two isoforms of Drosophila spectrin assemble independently of one another, and that ankyrin is upstream of alphabeta spectrin in the spectrin assembly pathway. In contrast, beta spectrin mutations had a striking effect on the basolateral accumulation of the Na,K ATPase. The results establish a role for beta spectrin in determining the subcellular distribution of the Na, K ATPase and, unexpectedly, this role is independent of alpha spectrin.
Figures




References
-
- Baumann O., Lautenschlager B., Takeyasu K. Immunolocalization of Na,K-ATPase in blowfly photoreceptor cells. Cell Tissue Res. 1994;275:225–234. - PubMed
-
- Bennett V., Gilligan D.M. The spectrin-based membrane skeleton and micron-scale organization of the plasma membrane. Annu. Rev. Cell Biol. 1993;9:27–66. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

