The role of Fcgamma receptor polymorphisms and C3 in the immune defence against Neisseria meningitidis in complement-deficient individuals
- PMID: 10792385
- PMCID: PMC1905639
- DOI: 10.1046/j.1365-2249.2000.01208.x
The role of Fcgamma receptor polymorphisms and C3 in the immune defence against Neisseria meningitidis in complement-deficient individuals
Abstract
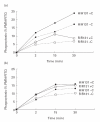
Individuals with either a late (C5-9) complement component deficiency (LCCD) or properdin deficiency are at increased risk to develop meningococcal disease, often due to serogroups W135 and Y. Anti-meningococcal defence in both LCCD persons and properdin-deficient individuals without bactericidal antibodies depends mainly on phagocytosis. Three types of opsonin receptors are involved in phagocytosis by polymorphonuclear cells (PMN). These represent the polymorphic FcgammaRIIa (CD32) and FcgammaRIIIb (CD16b) receptors, and the C3 receptor CR3 (CD11b/CD18). When the distribution of FcgammaRIIa and FcgammaRIIIb allotypes was assessed in 15 LCCD and in 15 properdin-deficient patients with/without previous meningococcal disease, we found the combination of FcgammaRIIa-R/R131 with FcgammaRIIIb-NA2/NA2 allotypes to be associated with previous meningococcal disease (odds ratio 13.9, Fisher's test P = 0.036). No such relation was observed in the properdin-deficient patients. The importance of FcgammaRIIa allotypes was also demonstrated using in vitro phagocytosis assays. PMN from FcgammaRIIa-R/R131 homozygous donors internalized IgG2 opsonized meningococci W135 significantly (P < 0.05) less than PMN from FcgammaRIIa-H/H131 donors. When properdin-deficient serum was tested, it was observed that reconstitution with properdin resulted in enhanced PMN phagocytosis of the W135 meningococci (P = 0.001). This enhanced phagocytosis was parallelled by an increase in C3 deposition onto the opsonized meningococci W135 (r = 0.6568, P = 0. 01). We conclude that the occurrence of meningococcal disease in LCCD patients is associated with certain FcgammaR allotypes. Properdin-deficient individuals are susceptible to meningococcal disease because of an insufficient C3 deposition on the surface of meningococci, resulting in insufficient phagocytosis.
Figures



Similar articles
-
Bench-to-bedside review: genetic influences on meningococcal disease.Crit Care. 2002 Feb;6(1):60-5. doi: 10.1186/cc1454. Epub 2001 Nov 26. Crit Care. 2002. PMID: 11940267 Free PMC article. Review.
-
Meningococcal disease and polymorphism of FcgammaRIIa (CD32) in late complement component-deficient individuals.Clin Exp Immunol. 1998 Jan;111(1):97-101. doi: 10.1046/j.1365-2249.1998.00484.x. Clin Exp Immunol. 1998. PMID: 9472667 Free PMC article.
-
Protection against meningococcal serogroup ACYW disease in complement-deficient individuals vaccinated with the tetravalent meningococcal capsular polysaccharide vaccine.Clin Exp Immunol. 1998 Dec;114(3):362-9. doi: 10.1046/j.1365-2249.1998.00738.x. Clin Exp Immunol. 1998. PMID: 9844044 Free PMC article. Clinical Trial.
-
The role of complement in anti-bacterial defence.Ann Ital Med Int. 1994 Jul-Sep;9(3):173-7. Ann Ital Med Int. 1994. PMID: 7946895 Clinical Trial.
-
Complement deficiency states and meningococcal disease.Immunol Res. 1993;12(3):295-311. doi: 10.1007/BF02918259. Immunol Res. 1993. PMID: 8288947 Review.
Cited by
-
Meningococcal Vaccinations.Infect Dis Ther. 2016 Jun;5(2):89-112. doi: 10.1007/s40121-016-0107-0. Epub 2016 Apr 16. Infect Dis Ther. 2016. PMID: 27086142 Free PMC article. Review.
-
Complement component C7 deficiency in a Spanish family.Clin Exp Immunol. 2003 Aug;133(2):240-6. doi: 10.1046/j.1365-2249.2003.02186.x. Clin Exp Immunol. 2003. PMID: 12869030 Free PMC article.
-
Bench-to-bedside review: genetic influences on meningococcal disease.Crit Care. 2002 Feb;6(1):60-5. doi: 10.1186/cc1454. Epub 2001 Nov 26. Crit Care. 2002. PMID: 11940267 Free PMC article. Review.
-
Two novel mutations in the C7 gene in a Korean patient with complement C7 deficiency.J Korean Med Sci. 2005 Apr;20(2):220-4. doi: 10.3346/jkms.2005.20.2.220. J Korean Med Sci. 2005. PMID: 15831990 Free PMC article.
-
Early cytokine release in response to live Borrelia burgdorferi Sensu Lato Spirochetes is largely complement independent.PLoS One. 2014 Sep 29;9(9):e108013. doi: 10.1371/journal.pone.0108013. eCollection 2014. PLoS One. 2014. PMID: 25265036 Free PMC article.
References
-
- Fijen CAP, Kuijper EJ, Hannema AJ, Sjöholm AG, van Putten JPM. Complement deficiencies in patients over ten years old with meningococcal disease due to uncommon serogroups. Lancet. 1989;ii:585–8. - PubMed
-
- Würzner R, Orren A, Lachmann PJ. Inherited deficiencies of the terminal components of human complement. Immunodef Rev. 1992;3:123–47. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

