Characterization and isolation of ductular cells coexpressing neural cell adhesion molecule and Bcl-2 from primary cholangiopathies and ductal plate malformations
- PMID: 10793072
- PMCID: PMC1876925
- DOI: 10.1016/S0002-9440(10)65032-8
Characterization and isolation of ductular cells coexpressing neural cell adhesion molecule and Bcl-2 from primary cholangiopathies and ductal plate malformations
Abstract
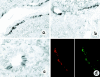
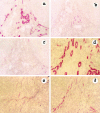
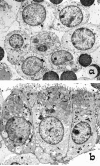
It has recently been shown that reactive bile ductules display neuroendocrine features, including immunoreactivity for the neural cell adhesion molecule (NCAM). In this study we have compared the immunohistochemical expression of NCAM with that of HEA-125 (biliary specific) and LKM-1 (hepatocyte specific) and other markers relevant to morphogenesis (Bcl-2, EMA) and cell proliferation (Ki-67) in cryostat sections from different chronic liver diseases and from fetal livers at different gestational ages. In parallel, viable NCAM-positive ductular cells were purified from collagenase digests of cirrhotic livers by immunomagnetic separation and characterized by immunocytochemistry and transmission electron microscopy. We demonstrated that reactive ductules with atypical morphology coexpressed NCAM and Bcl-2 and were found mainly in congenital diseases associated with ductal plate malformation and in primary cholangiopathies. On the contrary, reactive ductules with typical morphology were negative for NCAM/Bcl-2 and positive for EMA. Reactive ductules coexpressing NCAM/Bcl-2 were negative for the proliferation marker Ki-67 and appeared to be directly connected with periportal hepatocytes. In fetal livers NCAM/Bcl-2 was transiently expressed during the early developmental stages of ductal plate (10-16 weeks) and started to disappear as the ductal plate began duplicating. NCAM-positive ductal plate cells were Ki-67 negative, becoming positive in duplicated segments. Thus the histogenesis of ductular reactive cells seems to recapitulate the early stages of biliary ontogenesis. In primary cholangiopathies and ductal plate malformations, these cells do not appear to maturate further, and thus abundant ductular structures coexist with vanishing mature ducts. These NCAM-positive ductular cells were immunopurified from patients with chronic cholestatic liver diseases and showed ultrastructural features consistent with a less differentiated phenotype than mature cholangiocytes. These isolated cells represent a useful model for in vitro studies.
Figures


References
-
- Popper H, Schaffner F, Stein R: Ductular cell reaction in the liver in hepatic injury. J Mt Sinai Hosp 1957, 24:551-556 - PubMed
-
- Masuko K, Rubin E, Popper H: Proliferation of bile ducts in cirrhosis. Arch Pathol 1964, 78:421-431 - PubMed
-
- Desmet VJ, Roskams T, Van Eyken P: Ductular reaction in the liver. Pathol Res Pract 1995, 191:513-524 - PubMed
-
- Roskams T, Rosenbau J, De Vos R, David G, Desmet VJ: Heparan sulfate proteoglycan expression in chronic cholestatic human liver diseases. Hepatology 1996, 24:524-532 - PubMed
-
- Matsumoto K, Fuji H, Michalopoulos G, Fung JJ, Demetris AJ: Human biliary epithelial cells secrete and respond to cytokines and hepatocyte growth factors in vitro: interleukin-6, hepatocyte growth factor and epidermal growth factor promote DNA synthesis in vitro. Hepatology 1994, 20:376-382 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

