Neurodegeneration in Lurcher mice occurs via multiple cell death pathways
- PMID: 10804210
- PMCID: PMC6772704
- DOI: 10.1523/JNEUROSCI.20-10-03687.2000
Neurodegeneration in Lurcher mice occurs via multiple cell death pathways
Abstract
Lurcher (Lc) is a gain-of-function mutation in the delta2 glutamate receptor (GRID2) that results in the cell-autonomous death of cerebellar Purkinje cells in heterozygous lurcher (+/Lc) mice. This in turn triggers the massive loss of afferent granule cells during the first few postnatal weeks. Evidence suggests that the death of Purkinje cells as a direct consequence of GRID2(Lc) activation and the secondary death of granule cells because of target deprivation occur by apoptosis. We have used mice carrying null mutations of both the Bax and p53 genes to examine the roles of these genes in cell loss in lurcher animals. The absence of Bax delayed Purkinje cell death in response to the GRID2(Lc) mutation and permanently rescued the secondary death of granule cells. In contrast, the p53 deletion had no effect on either cell death pathway. Our results demonstrate that target deprivation induces a Bax-dependent, p53-independent cell death response in cerebellar granule cells in vivo. In contrast, Bax plays a minor role in GRID2(Lc)-mediated Purkinje cell death.
Figures
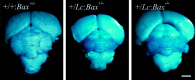

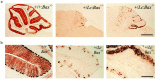

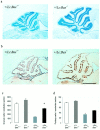
Similar articles
-
Bax inactivation in lurcher mutants rescues cerebellar granule cells but not purkinje cells or inferior olivary neurons.J Neurosci. 2000 Jul 15;20(14):5339-45. doi: 10.1523/JNEUROSCI.20-14-05339.2000. J Neurosci. 2000. PMID: 10884318 Free PMC article.
-
Bax and p53 are differentially involved in the regulation of caspase-3 expression and activation during neurodegeneration in Lurcher mice.C R Acad Sci III. 2000 Nov;323(11):967-73. doi: 10.1016/s0764-4469(00)01243-9. C R Acad Sci III. 2000. PMID: 11144029
-
Lurcher GRID2-induced death and depolarization can be dissociated in cerebellar Purkinje cells.Neuron. 2003 Mar 6;37(5):813-9. doi: 10.1016/s0896-6273(03)00093-x. Neuron. 2003. PMID: 12628171
-
Cell death, Bcl-2, Bax, and the cerebellum.Cerebellum. 2002 Dec;1(4):277-87. doi: 10.1080/147342202320883588. Cerebellum. 2002. PMID: 12879966 Review.
-
Mechanisms of compartmental purkinje cell death and survival in the lurcher mutant mouse.Cerebellum. 2011 Sep;10(3):504-14. doi: 10.1007/s12311-010-0231-4. Cerebellum. 2011. PMID: 21104177 Review.
Cited by
-
Motor learning of mice lacking cerebellar Purkinje cells.Front Neuroanat. 2013 Apr 23;7:4. doi: 10.3389/fnana.2013.00004. eCollection 2013. Front Neuroanat. 2013. PMID: 23630472 Free PMC article.
-
Smaller Absolute Quantities but Greater Relative Densities of Microvessels Are Associated with Cerebellar Degeneration in Lurcher Mice.Front Neuroanat. 2016 Apr 19;10:35. doi: 10.3389/fnana.2016.00035. eCollection 2016. Front Neuroanat. 2016. PMID: 27147979 Free PMC article.
-
Local insulin-like growth factor I expression is essential for Purkinje neuron survival at birth.Cell Death Differ. 2011 Jan;18(1):48-59. doi: 10.1038/cdd.2010.78. Epub 2010 Jul 2. Cell Death Differ. 2011. PMID: 20596079 Free PMC article.
-
Comparison of Cbln1 and Cbln2 functions using transgenic and knockout mice.J Neurochem. 2012 Feb;120(4):528-40. doi: 10.1111/j.1471-4159.2011.07604.x. J Neurochem. 2012. PMID: 22117778 Free PMC article.
-
Friedreich ataxia mouse models with progressive cerebellar and sensory ataxia reveal autophagic neurodegeneration in dorsal root ganglia.J Neurosci. 2004 Feb 25;24(8):1987-95. doi: 10.1523/JNEUROSCI.4549-03.2004. J Neurosci. 2004. PMID: 14985441 Free PMC article.
References
-
- Ankarcrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, Nicotera P. Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron. 1995;15:961–973. - PubMed
-
- Caddy KW, Biscoe TJ. Structural and quantitative studies on the normal C3H and Lurcher mutant mouse. Philos Trans R Soc Lond [Biol] 1979;287:167–201. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
