Class II phosphoinositide 3-kinases are downstream targets of activated polypeptide growth factor receptors
- PMID: 10805725
- PMCID: PMC85707
- DOI: 10.1128/MCB.20.11.3817-3830.2000
Class II phosphoinositide 3-kinases are downstream targets of activated polypeptide growth factor receptors
Abstract
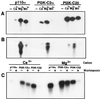
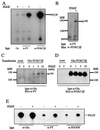
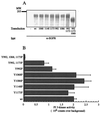
The class II phosphoinositide 3-kinases (PI3K) PI3K-C2alpha and PI3K-C2beta are two recently identified members of the large PI3K family. Both enzymes are characterized by the presence of a C2 domain at the carboxy terminus and, in vitro, preferentially utilize phosphatidylinositol and phosphatidylinositol 4-monophosphate as lipid substrates. Little is understood about how the catalytic activity of either enzyme is regulated in vivo. In this study, we demonstrate that PI3K-C2alpha and PI3K-C2beta represent two downstream targets of the activated epidermal growth factor (EGF) receptor in human carcinoma-derived A431 cells. Stimulation of quiescent cultures with EGF resulted in the rapid recruitment of both enzymes to a phosphotyrosine signaling complex that contained the EGF receptor and Erb-B2. Ligand addition also induced the appearance of a second, more slowly migrating band of PI3K-C2alpha and PI3K-C2beta immunoreactivity on sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Since both PI3K enzymes can utilize Ca(2+) as an essential divalent cation in lipid kinase assays and since the catalytic activity of PI3K-C2alpha is refractory to the inhibitor wortmannin, these properties were used to confirm the recruitment of each PI3K isozyme to the activated EGF receptor complex. To examine this interaction in greater detail, PI3K-C2beta was chosen for further investigation. EGF and platelet-derived growth factor also stimulated the association of PI3K-C2beta with their respective receptors in other cells, including epithelial cells and fibroblasts. The use of EGF receptor mutants and phosphopeptides derived from the EGF receptor and Erb-B2 demonstrated that the interaction with recombinant PI3K-C2beta occurs through E(p)YL/I phosphotyrosine motifs. The N-terminal region of PI3K-C2beta was found to selectively interact with the EGF receptor in vitro, suggesting that it mediates the association of this PI3K with the receptor. However, the mechanism of this interaction remains unclear. We conclude that class II PI3K enzymes may contribute to the generation of 3' phosphoinositides following the activation of polypeptide growth factor receptors in vivo and thus mediate certain aspects of their biological activity.
Figures







References
-
- Alessi D R, James S R, Downes C P, Holmes A B, Gaffney P R J, Reese C B, Cohen P. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Bα. Curr Biol. 1997;7:261–269. - PubMed
-
- Arcaro A, Volinia S, Zvelebil M J, Stein R, Watton S J, Layton M J, Gout I, Ahmadi K, Downward J, Waterfield M D. Human PI3-kinase C2β—the role of calcium and the C2 domain in enzyme activity. J Biol Chem. 1998;273:33082–33091. - PubMed
-
- Bondeva T, Pirola L, Bulgarelli-Leva G, Rubio I, Wetzker R, Wymann M. Bifurcation of lipid and protein kinase signals of PI3Kγ to the protein kinases PBK and MAPK. Science. 1998;282:293–296. - PubMed
-
- Brown R A, Domin J, Arcaro A, Waterfield M D, Shepherd P R. Insulin activates the alpha isoform of class II phosphoinositide 3-kinase. J Biol Chem. 1999;274:14529–14532. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
