Fruit-localized phytochromes regulate lycopene accumulation independently of ethylene production in tomato
- PMID: 10806253
- PMCID: PMC59010
- DOI: 10.1104/pp.123.1.363
Fruit-localized phytochromes regulate lycopene accumulation independently of ethylene production in tomato
Abstract
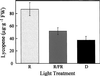
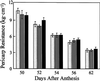
We show that phytochromes modulate differentially various facets of light-induced ripening of tomato fruit (Solanum lycopersicum L.). Northern analysis demonstrated that phytochrome A mRNA in fruit accumulates 11.4-fold during ripening. Spectroradiometric measurement of pericarp tissues revealed that the red to far-red ratio increases 4-fold in pericarp tissues during ripening from the immature-green to the red-ripe stage. Brief red-light treatment of harvested mature-green fruit stimulated lycopene accumulation 2. 3-fold during fruit development. This red-light-induced lycopene accumulation was reversed by subsequent treatment with far-red light, establishing that light-induced accumulation of lycopene in tomato is regulated by fruit-localized phytochromes. Red-light and red-light/far-red-light treatments during ripening did not influence ethylene production, indicating that the biosynthesis of this ripening hormone in these tissues is not regulated by fruit-localized phytochromes. Compression analysis of fruit treated with red light or red/far-red light indicated that phytochromes do not regulate the rate or extent of pericarp softening during ripening. Moreover, treatments with red or red/far-red light did not alter the concentrations of citrate, malate, fructose, glucose, or sucrose in fruit. These results are consistent with two conclusions: (a) fruit-localized phytochromes regulate light-induced lycopene accumulation independently of ethylene biosynthesis; and (b) fruit-localized phytochromes are not global regulators of ripening, but instead regulate one or more specific components of this developmental process.
Figures

References
-
- Bird CR, Ray JA, Fletcher JD, Boniwell JM, Bird AS, Teulieres C, Blain I, Bramley PM, Schuch W. Using antisense RNA to study gene function: inhibition of carotenoid biosynthesis in transgenic tomatoes. Biotechnology. 1991;9:635–639.
-
- Boe AA, Salunkhe DK. Ripening tomatoes: ethylene, oxygen, and light treatments. Econ Bot. 1967;21:321–319.
-
- Bushway RJ. Determination of α- and β-carotene in some raw fruits and vegetables by high-performance liquid chromatography. J Agric Food Chem. 1986;34:409–412.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

