Attenuation of pressure-induced myogenic contraction and tyrosine phosphorylation by fasudil, a cerebral vasodilator, in rat cerebral artery
- PMID: 10807658
- PMCID: PMC1572061
- DOI: 10.1038/sj.bjp.0703292
Attenuation of pressure-induced myogenic contraction and tyrosine phosphorylation by fasudil, a cerebral vasodilator, in rat cerebral artery
Abstract
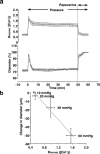
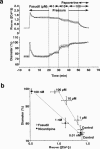
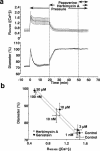
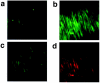
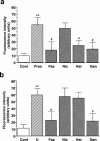
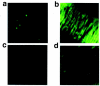
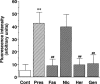
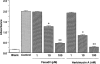
The mechanism by which fasudil inhibits pressure-induced myogenic contraction was studied with regard to tyrosine phosphorylation in rat cerebral artery. Intracellular Ca(2+) concentration ([Ca(2+)](i)) and vessel diameter were simultaneously measured. Total tyrosine phosphorylation level and phosphorylation of tyrosine 419 on pp60(src) required for its full catalytic activity were immunocytochemically detected in situ. Fasudil (1 - 100 microM) partially suppressed the increase in [Ca(2+)](i), and totally attenuated contraction elicited by pressurization from 10 to 60 mmHg. Furthermore, fasudil (100 microM) significantly attenuated tyrosine phosphorylation and the activity of pp60(src) augmented in situ by pressure. Herbimycin A (1 - 100 nM) and genistein (3 - 30 microM), tyrosine kinase inhibitors, effectively attenuated the pressure-induced increase in [Ca(2+)](i), contraction, tyrosine phosphorylation, and activation of pp60(src). Both fasudil and herbimycin A directly inhibited the pp60(src) activity in a cell free system. Orthovanadate (100 microM), a tyrosine phosphatase inhibitor, significantly potentiated the pressure-induced increase in [Ca(2+)](i) and contraction. Nicardipine (100 nM), a Ca(2+) antagonist, completely inhibited pressure-induced increase in [Ca(2+)](i) and contraction, but affected neither tyrosine phosphorylation nor activity of pp60(src) in the pressurized arteries. Arginine-glycine-aspartic acid-serine peptide (1 - 100 microM) concentration-dependently reduced the pressure-induced contraction. In addition to the hitherto reported vasodilatory actions of fasudil, the present results suggest the inhibition by fasudil of pressure-induced tyrosine phosphorylation and pp60(src) activation. The wide spectrum of inhibitory actions of fasudil may contribute to the effective attenuation of the pressure-induced contraction in the cerebral artery.
Figures
Similar articles
-
Role of pp60(c-src) and p(44/42) MAPK in ANG II-induced contraction of rat tonic gastrointestinal smooth muscles.Am J Physiol Gastrointest Liver Physiol. 2002 Aug;283(2):G390-9. doi: 10.1152/ajpgi.00025.2002. Am J Physiol Gastrointest Liver Physiol. 2002. PMID: 12121887
-
Specific attenuation of the pressure-induced contraction of rat cerebral artery by herbimycin A.Eur J Pharmacol. 1997 Jul 2;330(1):55-63. doi: 10.1016/s0014-2999(97)00166-0. Eur J Pharmacol. 1997. PMID: 9228414
-
Amiloride-sensitive pressure-induced myogenic contraction in rat cerebral artery.Fundam Clin Pharmacol. 2000 Jul-Aug;14(4):369-77. doi: 10.1111/j.1472-8206.2000.tb00418.x. Fundam Clin Pharmacol. 2000. PMID: 11030444
-
[The molecular mechanism of vasospasm and the attenuation by fasudil].Nihon Yakurigaku Zasshi. 1999 Oct;114 Suppl 1:66P-70P. doi: 10.1254/fpj.114.supplement_66. Nihon Yakurigaku Zasshi. 1999. PMID: 10629857 Review. Japanese.
-
[Fasudil hydrochloride].Nihon Rinsho. 2006 Oct 28;64 Suppl 7:617-21. Nihon Rinsho. 2006. PMID: 17461215 Review. Japanese. No abstract available.
Cited by
-
Vascular mechanotransduction.Physiol Rev. 2023 Apr 1;103(2):1247-1421. doi: 10.1152/physrev.00053.2021. Epub 2023 Jan 5. Physiol Rev. 2023. PMID: 36603156 Free PMC article. Review.
-
Rho kinase as a target for cerebral vascular disorders.Future Med Chem. 2015;7(8):1039-53. doi: 10.4155/fmc.15.45. Future Med Chem. 2015. PMID: 26062400 Free PMC article. Review.
-
Elucidating the impact of parthanatos-related microRNAs on the tumoral immune microenvironment and clinical outcome in low-grade gliomas.Discov Oncol. 2024 May 10;15(1):153. doi: 10.1007/s12672-024-01025-w. Discov Oncol. 2024. PMID: 38730061 Free PMC article.
References
-
- ASANO T., IKEGAKI I., SATOH S., SUZUKI Y., SHIBUYA M., TAKAYASU M., HIDAKA H. Mechanism of action of a novel antivasospasm drug, HA1077. J. Pharmacol. Exp. Ther. 1987;241:1033–1040. - PubMed
-
- D'ANGELO G., DAVIS M.J., MEININGER G.A. Calcium and mechanotransduction of the myogenic response. Am. J. Physiol. 1997a;273:H175–H182. - PubMed
-
- D'ANGELO G., MOGFORD J.E., DAVIS G.E., DAVIS M.J., MEININGER G.A. Integrin-mediated reduction in vascular smooth muscle [Ca2+]i induced by RGD-containing peptide. Am. J. Physiol. 1997b;272:H2065–H2070. - PubMed
-
- HIMPENS B., SMEDT H.D., DROOGMANS G., CASTEELS R. Differences in regulation between nuclear and cytoplasmic Ca2+ in cultured smooth muscle cells. Am. J. Physiol. 1992;263:C95–C105. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

