Yersinia enterocolitica TyeA, an intracellular regulator of the type III machinery, is required for specific targeting of YopE, YopH, YopM, and YopN into the cytosol of eukaryotic cells
- PMID: 10809698
- PMCID: PMC94505
- DOI: 10.1128/JB.182.11.3183-3190.2000
Yersinia enterocolitica TyeA, an intracellular regulator of the type III machinery, is required for specific targeting of YopE, YopH, YopM, and YopN into the cytosol of eukaryotic cells
Abstract
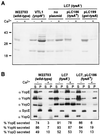
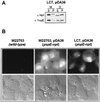
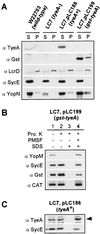
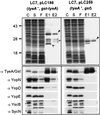
Pathogenic Yersinia species employ type III machines to target effector Yops into the cytosol of eukaryotic cells. Yersinia tyeA mutants are thought to be defective in the targeting of YopE and YopH without affecting the injection of YopM, YopN, YopO, YopP, and YopT into the cytosol of eukaryotic cells. One model suggests that TyeA may form a tether between YopN (LcrE) and YopD on the bacterial surface, a structure that may translocate YopE and YopH across the plasma membrane of eukaryotic cells (M. Iriarte, M. P. Sory, A. Boland, A. P. Boyd, S. D. Mills, I. Lambermont, and G. R. Cornelis, EMBO J. 17:1907-1918, 1998). We have examined the injection of Yop proteins by tyeA mutant yersiniae with the digitonin fractionation technique. We find that tyeA mutant yersiniae not only secreted YopE, YopH, YopM, and YopN into the extracellular medium but also targeted these polypeptides into the cytosol of HeLa cells. Protease protection, cell fractionation, and affinity purification experiments suggest that TyeA is located intracellularly and binds to YopN or YopD. We propose a model whereby TyeA functions as a negative regulator of the type III targeting pathway in the cytoplasm of yersiniae, presumably by preventing the export of YopN.
Figures


References
-
- Anderson D M, Schneewind O. A mRNA signal for the type III secretion of Yop proteins by Yersinia enterocolitica. Science. 1997;278:1140–1143. - PubMed
-
- Anderson D M, Schneewind O. Type III machines of Gram-negative pathogens: injecting virulence factors into host cells and more. Curr Opin Microbiol. 1999;2:18–24. - PubMed
-
- Anderson D M, Schneewind O. Yersinia enterocolitica type III secretion: an mRNA signal that couples translation and secretion of YopQ. Mol Microbiol. 1999;31:1139–1148. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

