Bruton's tyrosine kinase links the B cell receptor to nuclear factor kappaB activation
- PMID: 10811866
- PMCID: PMC2193152
- DOI: 10.1084/jem.191.10.1735
Bruton's tyrosine kinase links the B cell receptor to nuclear factor kappaB activation
Abstract
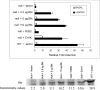
The recognition of antigen by membrane immunoglobulin M (mIgM) results in a complex series of signaling events in the cytoplasm leading to gene activation. Bruton's tyrosine kinase (BTK), a member of the Tec family of tyrosine kinases, is essential for the full repertoire of IgM signals to be transduced. We examined the ability of BTK to regulate the nuclear factor (NF)-kappaB/Rel family of transcription factors, as the activation of these factors is required for a B cell response to mIgM. We found greatly diminished IgM- but not CD40-mediated NF-kappaB/Rel nuclear translocation and DNA binding in B cells from X-linked immunodeficient (xid) mice that harbor an R28C mutation in btk, a mutation that produces a functionally inactive kinase. The defect was due, in part, to a failure to fully degrade the inhibitory protein of NF-kappaB, IkappaBalpha. Using a BTK-deficient variant of DT40 chicken B cells, we found that expression of wild-type or gain-of-function mutant BTK, but not the R28C mutant, reconstituted NF-kappaB activity. Thus, BTK is essential for activation of NF-kappaB via the B cell receptor.
Figures
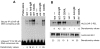



References
-
- Tsukada S., Saffran D.C., Rawlings D.J., Parolini O., Allen R.C., Klisak I., Sparkes R.S., Kubagawa H., Mohandas T., Quan S. Deficient expression of a B cell cytoplasmic tyrosine kinase in human X-linked agammaglobulinemia. Cell. 1993;72:279–290. - PubMed
-
- Rawlings D.J., Saffran D.C., Tsukada S., Largaespada D.A., Grimaldi J.C., Cohen L., Mohr R.N., Bazan J.F., Howard M., Copeland N.G. Mutation of unique region of Bruton's tyrosine kinase in immunodeficient XID mice. Science. 1993;261:358–361. - PubMed
-
- Thomas J.D., Sideras P., Smith C.I., Vorechovsky I., Chapman V., Paul W.E. Colocalization of X-linked agammaglobulinemia and X-linked immunodeficiency genes. Science. 1993;261:355–358. - PubMed
-
- Conley M.E., Parolini O., Rohrer J., Campana D. X-linked agammaglobulinemianew approaches to old questions based on the identification of the defective gene. Immunol. Rev. 1994;138:5–21. - PubMed
-
- Rawlings D.J., Witte O.N. Bruton's tyrosine kinase is a key regulator in B-cell development. Immunol. Rev. 1994;138:105–119. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

