Comparison of quantitative cytomegalovirus (CMV) PCR in plasma and CMV antigenemia assay: clinical utility of the prototype AMPLICOR CMV MONITOR test in transplant recipients
- PMID: 10834964
- PMCID: PMC86743
- DOI: 10.1128/JCM.38.6.2122-2127.2000
Comparison of quantitative cytomegalovirus (CMV) PCR in plasma and CMV antigenemia assay: clinical utility of the prototype AMPLICOR CMV MONITOR test in transplant recipients
Abstract
The correlation between the prototype AMPLICOR CMV MONITOR test (Roche Molecular Systems), a quantitative PCR assay, and the cytomegalovirus (CMV) pp65 antigenemia assay was evaluated in transplant recipients. Sequential blood specimens were collected on 29 patients (491 specimens), the leukocyte fraction was tested by CMV antigenemia, and quantitative PCR was performed on plasma specimens. None of the 15 patients (242 specimens) who were antigenemia negative were positive for CMV DNA by PCR, and none of these patients developed active CMV disease. There were 14 antigenemia-positive patients, 8 of whom developed active CMV disease. In all patients, there was a good association between the antigenemia and PCR assays. Ganciclovir-resistant virus was isolated from three patients with active CMV disease. These three patients had persistently elevated levels of antigenemia and CMV DNA by PCR when resistance to ganciclovir developed. This standardized, quantitative CMV PCR assay on plasma has clinical utility for the diagnosis of active disease and in monitoring the response to antiviral therapy in transplant recipients.
Figures

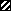 , intravitreal ganciclovir; ▤, intravenous foscarnet; ▥, oral
acyclovir; ■, intravenous ganciclovir; ▧, intravitreal foscarnet;
, intravitreal ganciclovir; ▤, intravenous foscarnet; ▥, oral
acyclovir; ■, intravenous ganciclovir; ▧, intravitreal foscarnet;
 ,
intravenous acyclovir;
,
intravenous acyclovir;
 ,
cytogam.
,
cytogam.
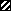 , intravitreal ganciclovir; ▤, intravenous foscarnet; ▥, oral
acyclovir; ■, intravenous ganciclovir; ▧, intravitreal foscarnet;
, intravitreal ganciclovir; ▤, intravenous foscarnet; ▥, oral
acyclovir; ■, intravenous ganciclovir; ▧, intravitreal foscarnet;
 ,
intravenous acyclovir;
,
intravenous acyclovir;
 ,
cytogam.
,
cytogam.References
-
- Abecassis M M, Koffron A J, Kaplan B, Buckingham M, Muldoon J P, Cribbins A J, Kaufman D B, Fryer J P, Stuart J, Stuart F P. The role of PCR in the diagnosis and management of CMV in solid organ transplant recipients. Transplantation. 1997;63:275–279. - PubMed
-
- Boeckh M, Gallez-Hawkins G M, Myerson D, Zaia J, Bowden R A. Plasma polymerase chain reaction for cytomegalovirus DNA after allogeneic bone marrow transplantation. Transplantation. 1997;64:108–113. - PubMed
-
- Brennan D C, Garlock K A, Lippmann B A, Buller R S, Gaudreault-Keener M, Lowell J A, Miller S B, Shenoy S, Howard T K, Storch G A. Control of cytomegalovirus-associated morbidity in renal transplant recipients using intensive monitoring and either preemptive or deferred therapy. J Am Soc Nephrol. 1997;8:118–125. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

