Herpes simplex virus type 1 entry into host cells: reconstitution of capsid binding and uncoating at the nuclear pore complex in vitro
- PMID: 10848617
- PMCID: PMC85943
- DOI: 10.1128/MCB.20.13.4922-4931.2000
Herpes simplex virus type 1 entry into host cells: reconstitution of capsid binding and uncoating at the nuclear pore complex in vitro
Abstract
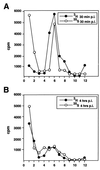
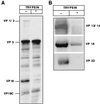
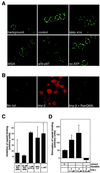
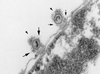
During entry, herpes simplex virus type 1 (HSV-1) releases its capsid and the tegument proteins into the cytosol of a host cell by fusing with the plasma membrane. The capsid is then transported to the nucleus, where it docks at the nuclear pore complexes (NPCs), and the viral genome is rapidly released into the nucleoplasm. In this study, capsid association with NPCs and uncoating of the viral DNA were reconstituted in vitro. Isolated capsids prepared from virus were incubated with cytosol and purified nuclei. They were found to bind to the nuclear pores. Binding could be inhibited by pretreating the nuclei with wheat germ agglutinin, anti-NPC antibodies, or antibodies against importin beta. Furthermore, in the absence of cytosol, purified importin beta was both sufficient and necessary to support efficient capsid binding to nuclei. Up to 60 to 70% of capsids interacting with rat liver nuclei in vitro released their DNA if cytosol and metabolic energy were supplied. Interaction of the capsid with the nuclear pore thus seemed to trigger the release of the viral genome, implying that components of the NPC play an active role in the nuclear events during HSV-1 entry into host cells.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
