The nonreceptor tyrosine kinase fer mediates cross-talk between N-cadherin and beta1-integrins
- PMID: 10851023
- PMCID: PMC2175119
- DOI: 10.1083/jcb.149.6.1263
The nonreceptor tyrosine kinase fer mediates cross-talk between N-cadherin and beta1-integrins
Abstract
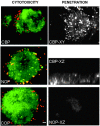
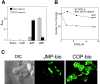
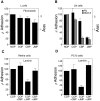
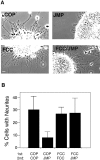
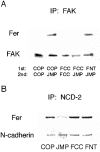
Cadherins and integrins must function in a coordinated manner to effectively mediate the cellular interactions essential for development. We hypothesized that exchange of proteins associated with their cytoplasmic domains may play a role in coordinating function. To test this idea, we used Trojan peptides to introduce into cells and tissues peptide sequences designed to compete for the interaction of specific effectors with the cytoplasmic domain of N-cadherin, and assayed their effect on cadherin- and integrin-mediated adhesion and neurite outgrowth. We show that a peptide mimicking the juxtamembrane (JMP) region of the cytoplasmic domain of N-cadherin results in inhibition of N-cadherin and beta1-integrin function. The effect of JMP on beta1-integrin function depends on the expression of N-cadherin and is independent of transcription or translation. Treatment of cells with JMP results in the release of the nonreceptor tyrosine kinase Fer from the cadherin complex and its accumulation in the integrin complex. A peptide that mimics the first coiled-coil domain of Fer prevents Fer accumulation in the integrin complex and reverses the inhibitory effect of JMP. These findings suggest a new mechanism through which N-cadherin and beta1-integrins are coordinately regulated: loss of an effector from the cytoplasmic domain of N-cadherin and gain of that effector by the beta1-integrin complex.
Figures

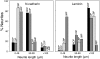
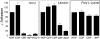


References
-
- Aberle H., Schwartz H., Kemler R. Cadherin catenin complexprotein interactions and their implications for cadherin function. J. Cell Biochem. 1996;61:514–523. - PubMed
-
- Aplin A.E., Howe A., Alahari S.K., Juliano R.L. Signal transduction and signal modulation by cell adhesion receptorsthe role of integrins, cadherins, immunoglobulin-cell adhesion molecules, and selectins. Pharm. Rev. 1998;50:199–263. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

