Coordinate regulation of cadherin and integrin function by the chondroitin sulfate proteoglycan neurocan
- PMID: 10851024
- PMCID: PMC2175121
- DOI: 10.1083/jcb.149.6.1275
Coordinate regulation of cadherin and integrin function by the chondroitin sulfate proteoglycan neurocan
Abstract
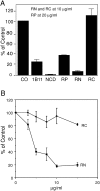
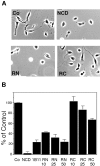
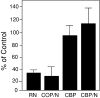
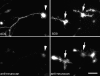
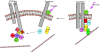
N-cadherin and beta1-integrins play decisive roles in morphogenesis and neurite extension and are often present on the same cell. Therefore, the function of these two types of adhesion systems must be coordinated in time and space to achieve the appropriate cell and tissue organization. We now show that interaction of the chondroitin sulfate proteoglycan neurocan with its GalNAcPTase receptor coordinately inhibits both N-cadherin- and beta1-integrin-mediated adhesion and neurite outgrowth. Furthermore, the inhibitory activity is localized to an NH(2)-terminal fragment of neurocan containing an Ig loop and an HA-binding domain. The effect of neurocan on beta1-integrin function is dependent on a signal originating from the cadherin cytoplasmic domain, possibly mediated by the nonreceptor protein tyrosine kinase Fer, indicating that cadherin and integrin engage in direct cross-talk. In the developing chick, neural retina neurocan is present in the inner plexiform layer from day 7 on, and the GalNAcPTase receptor becomes restricted to the inner nuclear layer and the ganglion cell layer (as well as the fiber layer), the two forming a sandwich. These data suggest that the coordinate inhibition of cadherin and integrin function on interaction of neurocan with its receptor may prevent cell and neurite migration across boundaries.
Figures

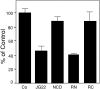
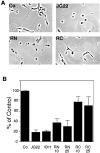
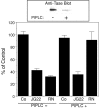

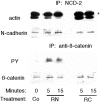
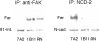


References
-
- Balsamo J., Lilien J. N-cadherin is stably associated with, and is an acceptor for, a cell surface N-acetylgalactosaminylphosphotransferase. J. Biol. Chem. 1990;265:2923–2928. - PubMed
-
- Balsamo J., Lilien J. The retina cell-surface N-acetylgalactosaminylphosphotransferase is anchored by a glycophosphatidylinositol. Biochemistry. 1993;32:8246–8250. - PubMed
-
- Balsamo J., Hoffman S., Lilien J. Control of cadherin-mediated cell-cell adhesion through regulated association with the cytoskeleton. J. Braz. Assoc. Adv. Sci. 1996;48:341–346.
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

