Regulation of cell invasion and morphogenesis in a three-dimensional type I collagen matrix by membrane-type matrix metalloproteinases 1, 2, and 3
- PMID: 10851027
- PMCID: PMC2175112
- DOI: 10.1083/jcb.149.6.1309
Regulation of cell invasion and morphogenesis in a three-dimensional type I collagen matrix by membrane-type matrix metalloproteinases 1, 2, and 3
Abstract
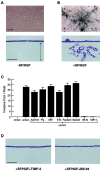
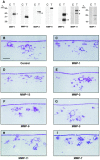
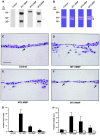
During tissue-invasive events, migrating cells penetrate type I collagen-rich interstitial tissues by mobilizing undefined proteolytic enzymes. To screen for members of the matrix metalloproteinase (MMP) family that mediate collagen-invasive activity, an in vitro model system was developed wherein MDCK cells were stably transfected to overexpress each of ten different MMPs that have been linked to matrix remodeling states. MDCK cells were then stimulated with scatter factor/hepatocyte growth factor (SF/HGF) to initiate invasion and tubulogenesis atop either type I collagen or interstitial stroma to determine the ability of MMPs to accelerate, modify, or disrupt morphogenic responses. Neither secreted collagenases (MMP-1 and MMP-13), gelatinases (gelatinase A or B), stromelysins (MMP-3 and MMP-11), or matrilysin (MMP-7) affected SF/HGF-induced responses. By contrast, the membrane-anchored metalloproteinases, membrane-type 1 MMP, membrane-type 2 MMP, and membrane-type 3 MMP (MT1-, MT2-, and MT3-MMP) each modified the morphogenic program. Of the three MT-MMPs tested, only MT1-MMP and MT2-MMP were able to directly confer invasion-incompetent cells with the ability to penetrate type I collagen matrices. MT-MMP-dependent invasion proceeded independently of proMMP-2 activation, but required the enzymes to be membrane-anchored to the cell surface. These findings demonstrate that MT-MMP-expressing cells can penetrate and remodel type I collagen-rich tissues by using membrane-anchored metalloproteinases as pericellular collagenases.
Figures




References
-
- Aimes R.T., Quigley J.P. Matrix metalloproteinase-2 is an interstitial collagenase. J. Biol. Chem. 1995;270:5872–5876. - PubMed
-
- Benbow U., Schoenermark M.P., Mitchell T.I., Rutter J.L., Shimokawa K., Nagase H., Brinckerhoff C.E. A novel host/tumor cell interaction activates matrix metalloproteinase 1 and mediates invasion through type I collagen. J. Biol. Chem. 1999;274:25371–25378. - PubMed
-
- Birchmeier C., Birchmeier W. Molecular aspects of mesenchymal–epithelial interactions. Annu. Rev. Cell Biol. 1993;9:511–540. - PubMed
-
- Birkedal-Hansen H. Catabolism and turnover of collagenscollagenases. Meth. Enzymol. 1987;144:140–167. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

