Biosynthesis of a major lipofuscin fluorophore in mice and humans with ABCR-mediated retinal and macular degeneration
- PMID: 10852960
- PMCID: PMC16515
- DOI: 10.1073/pnas.130110497
Biosynthesis of a major lipofuscin fluorophore in mice and humans with ABCR-mediated retinal and macular degeneration
Abstract
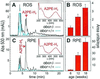
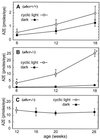
Increased accumulation of lipofuscin in cells of the retinal pigment epithelium (RPE) is seen in several forms of macular degeneration, a common cause of blindness in humans. A major fluorophore of lipofuscin is the toxic bis-retinoid, N-retinylidene-N-retinylethanolamine (A2E). Previously, we generated mice with a knockout mutation in the abcr gene. This gene encodes rim protein (RmP), an ATP-binding cassette transporter in rod outer segments. Mice lacking RmP accumulate A2E in RPE cells at a greatly increased rate over controls. Here, we identify three precursors of A2E in ocular tissues from abcr-/- mice and humans with ABCR-mediated recessive macular degenerations. Our results corroborate the scheme proposed by C. A. Parish, M. Hashimoto, K. Nakanishi, J. Dillon & J. Sparrow [Proc. Natl. Acad. Sci. USA (1998) 95, 14609-14613], for the biosynthesis of A2E: (i) condensation of all-trans-retinaldehyde (all-trans-RAL) with phosphatidylethanolamine to form a Schiff base; (ii) condensation of the amine product with a second all-trans-RAL to form a bis-retinoid; (iii) oxidation to yield a pyridinium salt; and (iv) hydrolysis of the phosphate ester to yield A2E. The latter two reactions probably occur within RPE phagolysosomes. As predicted by this model, formation of A2E was completely inhibited when abcr-/- mice were raised in total darkness. Also, once formed, A2E was not eliminated by the RPE. These data suggest that humans with retinal or macular degeneration caused by loss of RmP function may slow progression of their disease by limiting exposure to light. The precursors of A2E identified in this study may represent pharmacological targets for the treatment of ABCR-mediated macular degeneration.
Figures



References
-
- Bressler N M, Bressler S B, Fine S L. Surv Ophthalmol. 1988;32:375–413. - PubMed
-
- Bressler N M, Silva J C, Bressler S B, Fine S L, Green W R. Retina. 1994;14:130–142. - PubMed
-
- Kliffen M, van der Schaft T L, Mooy C M, de Jong P T. Microsc Res Tech. 1997;36:106–122. - PubMed
-
- Delori F C, Fleckner M R, Goger D G, Weiter J J, Dorey C K. Invest Ophthalmol Visual Sci. 2000;41:496–504. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

