Induction of HHV-8 lytic cycle replication by inflammatory cytokines produced by HIV-1-infected T cells
- PMID: 10854219
- PMCID: PMC1850066
- DOI: 10.1016/S0002-9440(10)65069-9
Induction of HHV-8 lytic cycle replication by inflammatory cytokines produced by HIV-1-infected T cells
Abstract
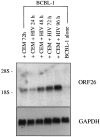
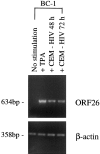
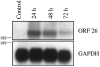
Human herpesvirus 8 (HHV-8) is a gamma2-herpesvirus consistently identified in Kaposi's sarcoma (KS), primary effusion lymphoma, and multicentric Castleman's disease. Although HHV-8 infection appears to be necessary, it may not be sufficient for development of KS without the involvement of other cofactors. One potentially important cofactor is HIV-1. HIV-1-infected cells produce HIV-1-related proteins and cytokines, both of which have been shown to promote growth of KS cells in vitro. Though HIV-1 is not absolutely necessary for KS development, KS is the most frequent neoplasm in AIDS patients, and AIDS-KS is recognized as a particularly aggressive form of the disease. To determine whether HIV-1 could participate in the pathogenesis of KS by modulating HHV-8 replication (rather than by inducing immunodeficiency), HIV-1-infected T cells were cocultured with the HHV-8-infected cell line, BCBL-1. The results demonstrate soluble factors produced by or in response to HIV-1-infected T cells induced HHV-8 replication, as determined by production of lytic phase mRNA transcripts, viral proteins, and detection of progeny virions. By focusing on cytokines produced in the coculture system, several cytokines known to be important in growth and proliferation of KS cells in vitro, particularly Oncostatin M, hepatocyte growth factor/scatter factor, and interferon-gamma, were found to induce HHV-8 lytic replication when added individually to BCBL-1 cells. These results suggest specific cytokines can play an important role in the initiation and progression of KS through reactivation of HHV-8. Thus, HIV-1 may participate more directly than previously recognized in KS by promoting HHV-8 replication and, hence, increasing local HHV-8 viral load.
Figures



Similar articles
-
Human herpesvirus 6 activates lytic cycle replication of Kaposi's sarcoma-associated herpesvirus.Am J Pathol. 2005 Jan;166(1):173-83. doi: 10.1016/S0002-9440(10)62242-0. Am J Pathol. 2005. PMID: 15632010 Free PMC article.
-
Human herpesvirus 8 reactivation and human immunodeficiency virus type 1 gp120.Arch Pathol Lab Med. 2002 Aug;126(8):941-6. doi: 10.5858/2002-126-0941-HHRAHI. Arch Pathol Lab Med. 2002. PMID: 12171492
-
Human herpesvirus 8 induces polyfunctional B lymphocytes that drive Kaposi's sarcoma.mBio. 2014 Sep 2;5(5):e01277-14. doi: 10.1128/mBio.01277-14. mBio. 2014. PMID: 25182322 Free PMC article.
-
Some aspects of the pathogenesis of HIV-1-associated Kaposi's sarcoma.J Natl Cancer Inst Monogr. 1998;(23):55-7. doi: 10.1093/oxfordjournals.jncimonographs.a024174. J Natl Cancer Inst Monogr. 1998. PMID: 9709304 Review.
-
Human herpesvirus 8 virology, epidemiology and related diseases.Jpn J Infect Dis. 2000 Aug;53(4):137-55. Jpn J Infect Dis. 2000. PMID: 11056556 Review.
Cited by
-
Cytokine-Targeted Therapeutics for KSHV-Associated Disease.Viruses. 2020 Sep 28;12(10):1097. doi: 10.3390/v12101097. Viruses. 2020. PMID: 32998419 Free PMC article. Review.
-
Molecular genetics of Kaposi's sarcoma-associated herpesvirus (human herpesvirus-8) epidemiology and pathogenesis.Microbiol Mol Biol Rev. 2003 Jun;67(2):175-212, table of contents. doi: 10.1128/MMBR.67.2.175-212.2003. Microbiol Mol Biol Rev. 2003. PMID: 12794189 Free PMC article. Review.
-
Molecular biology of KSHV in relation to AIDS-associated oncogenesis.Cancer Treat Res. 2007;133:69-127. doi: 10.1007/978-0-387-46816-7_3. Cancer Treat Res. 2007. PMID: 17672038 Free PMC article. Review.
-
Association between KSHV-Specific Humoral and T Cell Responses with Recurrence of HIV-Associated Kaposi Sarcoma.Trop Med Infect Dis. 2024 Jun 18;9(6):134. doi: 10.3390/tropicalmed9060134. Trop Med Infect Dis. 2024. PMID: 38922046 Free PMC article.
-
Host-Level Susceptibility and IRF1 Expression Influence the Ability of IFN-γ to Inhibit KSHV Infection in B Lymphocytes.Viruses. 2022 Oct 19;14(10):2295. doi: 10.3390/v14102295. Viruses. 2022. PMID: 36298850 Free PMC article.
References
-
- Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM, Moore PS: Identification of herpesvirus-like DNA sequences in AIDS- associated Kaposi’s sarcoma. Science 1994, 266:1865-1869 - PubMed
-
- Schulz TF: Kaposi’s sarcoma-associated herpesvirus (human herpesvirus-8). J Gen Virol 1998, 79:1573-1591 - PubMed
-
- Huang YQ, Li JJ, Kaplan MH, Poiesz B, Katabira E, Zhang WC, Feiner D, Friedman-Kien AE: Human herpesvirus-like nucleic acid in various forms of Kaposi’s sarcoma. Lancet 1995, 345:759-761 - PubMed
-
- Moore PS, Chang Y: Detection of herpesvirus-like DNA sequences in Kaposi’s sarcoma in patients with and without HIV infection. N Engl J Med 1995, 332:1181-1185 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

