The in vitro differentiating capacity of nonparenchymal epithelial cells derived from adult porcine livers
- PMID: 10854225
- PMCID: PMC1850063
- DOI: 10.1016/S0002-9440(10)65075-4
The in vitro differentiating capacity of nonparenchymal epithelial cells derived from adult porcine livers
Abstract
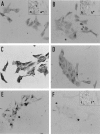
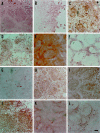
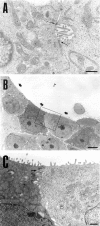
Specific nonparenchymal epithelial cell (NPEC) clusters derived from normal adult porcine livers demonstrate a characteristic developmental pattern in the presence of other types of nonparenchymal cells in vitro. This pattern includes scattering, colonial growth, and an emergence of duct-like structures (DLSs) in the colonies. It has been confirmed that 96% of the scattered cell clusters in these cultures develop into colonies containing DLSs. In this study, we examine the differentiation of NPEC clusters using the scattered formation as a marker of the DLS-emerged colonies. We report that the NPECs expressed albumin, alpha-fetoprotein, transferrin, cytokeratin (CK) 18, CK7, and c-met, but not alpha-1-antitrypsin (AAT), at the scattering stage. In addition, at the same stage, NPECs expressed oval-cell-related markers such as OV6, but not biliary epithelial cell (BEC) markers such as gamma-glutamyltransferase, CK19, and CK14. At the DLS emerging stage, hepatocyte markers, including AAT, were detectable in the cells either at the periphery of colonies or in the cells surrounded by the DLSs. On the other hand, the cells constituting DLSs expressed BEC markers, suggesting a bile duct nature of the DLSs. Furthermore, the cells in the colonies possessed an ultrastructural appearance of differentiated hepatocytes and BECs. These results suggest that certain NPECs are bipotent, and that, in culture, they mimic hepatoblast development in vivo.
Figures



Similar articles
-
Colony-forming capacity of porcine liver epithelial cells in culture.In Vitro Cell Dev Biol Anim. 2002 Sep;38(8):440-2. doi: 10.1290/1071-2690(2002)038<0440:CFCOPL>2.0.CO;2. In Vitro Cell Dev Biol Anim. 2002. PMID: 12605537
-
Colonial growth and differentiation of epithelial cells derived from abattoir adult porcine livers.J Gastroenterol Hepatol. 1998 Sep;13 Suppl:S62-9. J Gastroenterol Hepatol. 1998. PMID: 9792036
-
Identification of bipotential progenitor cells in human liver development.Hepatology. 1996 Mar;23(3):476-81. doi: 10.1002/hep.510230312. Hepatology. 1996. PMID: 8617427
-
Development and molecular composition of the hepatic progenitor cell niche.Dan Med J. 2013 May;60(5):B4640. Dan Med J. 2013. PMID: 23673270 Review.
-
Ductular hepatocytes.Histol Histopathol. 1995 Apr;10(2):433-56. Histol Histopathol. 1995. PMID: 7599440 Review.
Cited by
-
Colony-forming capacity of porcine liver epithelial cells in culture.In Vitro Cell Dev Biol Anim. 2002 Sep;38(8):440-2. doi: 10.1290/1071-2690(2002)038<0440:CFCOPL>2.0.CO;2. In Vitro Cell Dev Biol Anim. 2002. PMID: 12605537
-
Presence of side-population cells in an immortalized nontumorigenic human liver epithelial cell line.In Vitro Cell Dev Biol Anim. 2008 Jan-Feb;44(1-2):6-9. doi: 10.1007/s11626-007-9071-z. Epub 2007 Dec 22. In Vitro Cell Dev Biol Anim. 2008. PMID: 18157683
-
Hepatic stem cells: from inside and outside the liver?Cell Prolif. 2004 Feb;37(1):1-21. doi: 10.1111/j.1365-2184.2004.00297.x. Cell Prolif. 2004. PMID: 14871234 Free PMC article. Review.
-
Hepatic reconstruction from fetal porcine liver cells using a radial flow bioreactor.World J Gastroenterol. 2008 May 7;14(17):2740-7. doi: 10.3748/wjg.14.2740. World J Gastroenterol. 2008. PMID: 18461659 Free PMC article.
-
Side Population Cells From an Immortalized Human Liver Epithelial Cell Line Exhibit Hepatic Stem-Like Cell Properties.Cell Med. 2012 Jun 15;3(1-3):127-135. doi: 10.3727/215517912X639450. eCollection 2012 Jan. Cell Med. 2012. PMID: 28058190 Free PMC article.
References
-
- Shiojiri N, Lemire JM, Fausto N: Cell lineages and oval cell progenitors in rat liver development. Cancer Res 1991, 51:2611-2620 - PubMed
-
- Germain L, Blouin M-J, Marceau N: Biliary epithelial and hepatocytic cell lineage relationships in embryonic rat liver as determined by the differential expression of cytokeratins, α-fetoprotein, albumin, and cell surface-exposed components. Cancer Res 1988, 48:4909-4918 - PubMed
-
- Marceau N, Chamberland C, Blouin M-J, Noel M, Loranger A: Progenitor cells in embryonic and post-natal rat livers, their growth and differentiation potential. Bell E eds. Tissue Engineering Current Perspectives. 1993, :pp 58-65 Birkhauser, Berlin
-
- Grisham JW: Cell types in rat liver cultures: their identification and isolation. Mol Cell Biochem 1983, 53/54:23-33 - PubMed
-
- Tokiwa T, Nakabayashi H, Miyazaki M, Sato J: Isolation and characterization of diploid clones from adult and newborn rat liver cell lines. In Vitro 1979, 15:393-400 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

