Acid potentiation of the capsaicin receptor determined by a key extracellular site
- PMID: 10859346
- PMCID: PMC16682
- DOI: 10.1073/pnas.100129497
Acid potentiation of the capsaicin receptor determined by a key extracellular site
Abstract
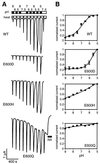
The capsaicin (vanilloid) receptor, VR1, is a sensory neuron-specific ion channel that serves as a polymodal detector of pain-producing chemical and physical stimuli. The response of VR1 to capsaicin or noxious heat is dynamically potentiated by extracellular protons within a pH range encountered during tissue acidosis, such as that associated with arthritis, infarction, tumor growth, and other forms of injury. A molecular determinant for this important physiological activity was localized to an extracellular Glu residue (E600) in the region linking the fifth transmembrane domain with the putative pore-forming region of the channel. We suggest that this residue serves as a key regulatory site of the receptor by setting sensitivity to other noxious stimuli in response to changes in extracellular proton concentration. We also demonstrate that protons, vanilloids, and heat promote channel opening through distinct pathways, because mutations at a second site (E648) selectively abrogate proton-evoked channel activation without diminishing responses to other noxious stimuli. Our findings provide molecular evidence for stimulus-specific steps in VR1 activation and offer strategies for the development of novel analgesic agents.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

