Tumour necrosis factor-alpha- and interleukin-1beta-stimulated cell proliferation through activation of mitogen-activated protein kinase in canine tracheal smooth muscle cells
- PMID: 10864897
- PMCID: PMC1572122
- DOI: 10.1038/sj.bjp.0703359
Tumour necrosis factor-alpha- and interleukin-1beta-stimulated cell proliferation through activation of mitogen-activated protein kinase in canine tracheal smooth muscle cells
Abstract
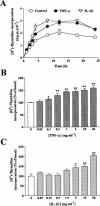
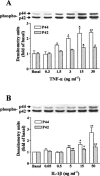
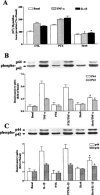
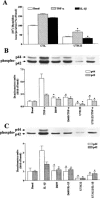
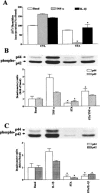
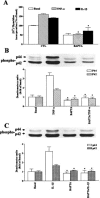
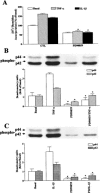
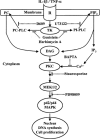
The elevated levels of inflammatory cytokines such as tumour necrosis factor-alpha (TNF-alpha) and interleukin-1beta (IL-1beta) have been found in the fluid of airways in symptomatic asthmatics. These cytokines have been considered as mitogens to stimulate cell proliferation in tracheal smooth muscle cells (TSMCs). We therefore investigated the effects of TNF-alpha and IL-1beta on cell proliferation and activation of p42/p44 mitogen-activated protein kinase (MAPK) in these cells. TNF-alpha and IL-1beta induced [(3)H]-thymidine incorporation in a time- and concentration-dependent manner. The maximal stimulation of [(3)H]-thymidine incorporation induced by TNF-alpha and IL-1beta was seen 12 h after incubation with these cytokines. In response to TNF-alpha and IL-1beta, p42/p44 MAPK was activated with a concentration-dependent manner in TSMCs. Pretreatment of TSMCs with pertussis toxin did not change DNA synthesis and phosphorylation of MAPK induced by TNF-alpha and IL-1beta. These responses were attenuated by a tyrosine kinase inhibitor herbimycin, a phosphatidyl choline (PC)-phospholipase C (PLC) inhibitor D609, a phosphatidyl inositide (PI)-PLC inhibitor U73122, a protein kinase C inhibitor staurosporine, and removal of Ca(2+) by addition of BAPTA/AM plus EGTA. TNF-alpha- and IL-1beta-induced [(3)H]-thymidine incorporation and phosphorylation of p42/p44 MAPK was completely inhibited by PD98059 (an inhibitor of MEK1/2), indicating that activation of MEK1/2 was required for these responses. These results suggest that the mitogenic effects of TNF-alpha and IL-1beta were mediated through the activation of MEK1/2 and p42/p44 MAPK pathway. TNF-alpha- and IL-1beta-mediated responses were modulated by PLC, Ca(2+), PKC, and tyrosine kinase associated with cell proliferation in TSMCs.
Figures
References
-
- ALESSI D.R., CUENDA A., COHEN P., DUDLEY D.T. , SATIEL A.R. PD098059 is a specific inhibitor of the activation of mitogen-activated protein kinase kinase in vitro and in vivo. J. Biol. Chem. 1995;270:27489–27494. - PubMed
-
- AMRANI Y., PANETTIERI R.A., FROSSARD N. , BRONNER C. Activation of the TNFα-p55 receptor induces myocyte proliferation and modulates agonist-evoked calcium transients in cultured human tracheal smooth muscle cells. Am. J. Respir. Cell Mol. Biol. 1996;15:55–63. - PubMed
-
- ARMOUR C.L., DIMENT L.M. , BLACK J.L. Relationship between smooth muscle volume and contractile response in airway tissue. Isometric versus isotonic measurement. J. Pharmacol. Exp. Ther. 1988;245:687–691. - PubMed
-
- BROIDE D.H., LOTZ M., CUOMO A.J., COBURN D.A., FEDERMAN E.C. , WASSERMAN S.I. Cytokines in symptomatic asthma airways. J. Allergy Clin. Immunol. 1992;89:958–967. - PubMed
-
- CEMBRZYNSKA-NOWAK M., SZKLARZ E., INGLOT A.D. , TEODORCZYK-INJEYAN J.A. Elevated release of tumor necrosis factor-alpha and interferone-gamma by bronchoalveolar leukocytes from patients with bronchial asthma. Am. Rev. Respir. Dis. 1993;147:291–295. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

