Coexpression of rat P2X2 and P2X6 subunits in Xenopus oocytes
- PMID: 10864944
- PMCID: PMC6772291
- DOI: 10.1523/JNEUROSCI.20-13-04871.2000
Coexpression of rat P2X2 and P2X6 subunits in Xenopus oocytes
Abstract
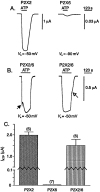
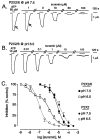
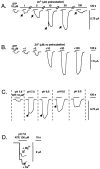
Transcripts for P2X(2) and P2X(6) subunits are present in rat CNS and frequently colocalize in the same brainstem nuclei. When rat P2X(2) (rP2X(2)) and rat P2X(6) (rP2X(6)) receptors were expressed individually in Xenopus oocytes and studied under voltage-clamp conditions, only homomeric rP2X(2) receptors were fully functional and gave rise to large inward currents (2-3 microA) to extracellular ATP. Coexpression of rP2X(2) and rP2X(6) subunits in Xenopus oocytes resulted in a heteromeric rP2X(2/6) receptor, which showed a significantly different phenotype from the wild-type rP2X(2) receptor. Differences included reduction in agonist potencies and, in some cases (e.g., Ap(4)A), significant loss of agonist activity. ATP-evoked inward currents were biphasic at the heteromeric rP2X(2/6) receptor, particularly when Zn(2+) ions were present or extracellular pH was lowered. The pH range was narrower for H(+) enhancement of ATP responses at the heteromeric rP2X(2/6) receptor. Also, H(+) ions inhibited ATP responses at low pH levels (<pH 6.3). The pH-dependent blocking activity of suramin was changed at this heteromeric receptor, although the potentiating effect of Zn(2+) on ATP responses was unchanged. Thus, the rP2X(2/6) receptor is a functionally modified P2X(2)-like receptor with a distinct pattern of pH modulation of ATP activation and suramin blockade. Although homomeric P2X(6) receptors function poorly, the P2X(6) subunit can contribute to functional heteromeric P2X channels and may influence the phenotype of native P2X receptors in those cells in which it is expressed.
Figures


References
-
- Barnard EA, Skolnick P, Olsen RW, Mohler H, Sieghart W, Biggio G, Braestrup C, Bateson AN, Langer SZ. International Union of Pharmacology. XV. Subtypes of gamma-aminobutyric acid (GABAA) receptors: classification on the basis of subunit structure and receptor function. Pharmacol Rev. 1998;50:291–313. - PubMed
-
- Buell G, Collo G, Rassendren F. P2X receptors: an emerging channel family. Eur J Neurosci. 1996;8:2221–2228. - PubMed
-
- Burnstock G. The past, present and future of purine nucleotides as signalling molecules. Neuropharmacology. 1997;36:1127–1139. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
