Phosphorylation of connexin43 on serine368 by protein kinase C regulates gap junctional communication
- PMID: 10871288
- PMCID: PMC2175134
- DOI: 10.1083/jcb.149.7.1503
Phosphorylation of connexin43 on serine368 by protein kinase C regulates gap junctional communication
Abstract
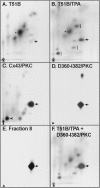
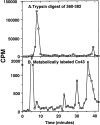
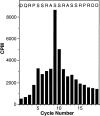
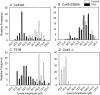
Phorbol esters (e.g., TPA) activate protein kinase C (PKC), increase connexin43 (Cx43) phosphorylation, and decrease cell-cell communication via gap junctions in many cell types. We asked whether PKC directly phosphorylates and regulates Cx43. Rat epithelial T51B cells metabolically labeled with (32)P(i) yielded two-dimensional phosphotryptic maps of Cx43 with several phosphopeptides that increased in intensity upon TPA treatment. One of these peptides comigrated with the major phosphopeptide observed after PKC phosphorylation of immunoaffinity-purified Cx43. Purification of this comigrating peptide and subsequent sequencing indicated that the phosphorylated serine was residue 368. To pursue the functional importance of phosphorylation at this site, fibroblasts from Cx43(-/-) mice were transfected with either wild-type (Cx43wt) or mutant Cx43 (Cx43-S368A). Intercellular dye transfer studies revealed different responses to TPA and were followed by single channel analyses. TPA stimulation of T51B cells or Cx43wt-transfected fibroblasts caused a large increase in the relative frequency of approximately 50-pS channel events and a concomitant loss of approximately 100-pS channel events. This change to approximately 50-pS events was absent when cells transfected with Cx43-S368A were treated with TPA. These data strongly suggest that PKC directly phosphorylates Cx43 on S368 in vivo, which results in a change in single channel behavior that contributes to a decrease in intercellular communication.
Figures


Similar articles
-
Connexin43 phosphorylation at S368 is acute during S and G2/M and in response to protein kinase C activation.J Cell Sci. 2003 Jun 1;116(Pt 11):2203-11. doi: 10.1242/jcs.00428. Epub 2003 Apr 15. J Cell Sci. 2003. PMID: 12697837
-
Epidermal growth factor stimulates the disruption of gap junctional communication and connexin43 phosphorylation independent of 12-0-tetradecanoylphorbol 13-acetate-sensitive protein kinase C: the possible involvement of mitogen-activated protein kinase.Mol Biol Cell. 1993 Aug;4(8):837-48. doi: 10.1091/mbc.4.8.837. Mol Biol Cell. 1993. PMID: 8241569 Free PMC article.
-
Phosphorylation of connexin43 and inhibition of gap junctional communication in 12-O-tetradecanoylphorbol-13-acetate-exposed R6 fibroblasts: minor role of protein kinase C beta I and mu.Carcinogenesis. 2001 Feb;22(2):221-31. doi: 10.1093/carcin/22.2.221. Carcinogenesis. 2001. PMID: 11181442
-
Connexin phosphorylation as a regulatory event linked to gap junction channel assembly.Biochim Biophys Acta. 2005 Jun 10;1711(2):154-63. doi: 10.1016/j.bbamem.2004.09.013. Epub 2004 Oct 12. Biochim Biophys Acta. 2005. PMID: 15955300 Review.
-
Phosphorylation of connexin43 induced by Src: regulation of gap junctional communication between transformed cells.Exp Cell Res. 2007 Dec 10;313(20):4083-90. doi: 10.1016/j.yexcr.2007.09.010. Epub 2007 Sep 20. Exp Cell Res. 2007. PMID: 17956757 Review.
Cited by
-
Mitotic cells form actin-based bridges with adjacent cells to provide intercellular communication during rounding.Cell Cycle. 2016 Nov;15(21):2943-2957. doi: 10.1080/15384101.2016.1231280. Epub 2016 Sep 13. Cell Cycle. 2016. PMID: 27625181 Free PMC article.
-
Molecular and Electrophysiological Mechanisms Underlying Cardiac Arrhythmogenesis in Diabetes Mellitus.J Diabetes Res. 2016;2016:2848759. doi: 10.1155/2016/2848759. Epub 2016 Aug 23. J Diabetes Res. 2016. PMID: 27642609 Free PMC article. Review.
-
Connexin43 Forms Supramolecular Complexes through Non-Overlapping Binding Sites for Drebrin, Tubulin, and ZO-1.PLoS One. 2016 Jun 9;11(6):e0157073. doi: 10.1371/journal.pone.0157073. eCollection 2016. PLoS One. 2016. PMID: 27280719 Free PMC article.
-
Levonorgestrel Inhibits Human Endometrial Cell Proliferation through the Upregulation of Gap Junctional Intercellular Communication via the Nuclear Translocation of Ser255 Phosphorylated Cx43.Biomed Res Int. 2015;2015:758684. doi: 10.1155/2015/758684. Epub 2015 Jun 16. Biomed Res Int. 2015. PMID: 26161412 Free PMC article.
-
PKCμ promotes keratinocyte cell migration through Cx43 phosphorylation-mediated suppression of intercellular communication.iScience. 2024 Jan 29;27(3):109033. doi: 10.1016/j.isci.2024.109033. eCollection 2024 Mar 15. iScience. 2024. PMID: 38375220 Free PMC article.
References
-
- Azarnia R., Loewenstein W.R. Intercellular communication and control of cell growth. X. Alteration of junctional permeability by the src gene. J. Membr. Biol. 1984;82:191–205. - PubMed
-
- Bazzi M.D., Nelsestuen G.L. Association of protein kinase C with phospholipid vesicles. Biochemistry. 1987;26:115–122. - PubMed
-
- Bazzi M.D., Nelsestuen G.L. Autophosphorylation of protein kinase C may require a high order of protein-phospholipid aggregates. J. Biol. Chem. 1992;267:22891–22896. - PubMed
-
- Berthoud V.M., Ledbetter M.L.S., Hertzberg E.L., Saez J.C. Connexin43 in MDCK cellsregulation by a tumor-promoting phorbol ester and calcium. Eur. J. Cell Biol. 1992;57:40–50. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

