Isolation and characterization of Tn7 transposase gain-of-function mutants: a model for transposase activation
- PMID: 10880457
- PMCID: PMC313929
- DOI: 10.1093/emboj/19.13.3446
Isolation and characterization of Tn7 transposase gain-of-function mutants: a model for transposase activation
Abstract
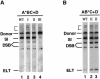
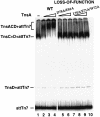
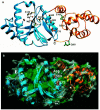
Tn7 transposition has been hypothesized to require a heteromeric transposase formed by two Tn7-encoded proteins, TnsA and TnsB, and accessory proteins that activate the transposase when they are associated with an appropriate target DNA. This study investigates the mechanism of Tn7 transposase activation by isolation and analysis of transposase gain-of-function mutants that are active in the absence of these accessory proteins. This work shows directly that TnsA and TnsB are essential and sufficient components of the Tn7 transposase and also provides insight into the signals that activate the transposase. We also describe a protein-protein interaction between TnsA and TnsC, a regulatory accessory protein, that is likely to be critical for transposase activation.
Figures

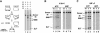



References
-
- Arciszewska L.K., McKown,R.L. and Craig,N.L. (1991) Purification of TnsB, a transposition protein that binds to the ends of Tn7. J. Biol. Chem., 266, 21736–21744. - PubMed
-
- Bainton R., Gamas,P. and Craig,N.L. (1991) Tn7 transposition in vitro proceeds through an excised transposon intermediate generated by staggered breaks in DNA. Cell, 65, 805–816. - PubMed
-
- Bainton R.J., Kubo,K.M., Feng,J.-N. and Craig,N.L. (1993) Tn7 transposition: target DNA recognition is mediated by multiple Tn7-encoded proteins in a purified in vitro system. Cell, 72, 931–943. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

