Differential regulation of nitric oxide synthase isoforms in experimental acute chagasic cardiomyopathy
- PMID: 10886247
- PMCID: PMC1905681
- DOI: 10.1046/j.1365-2249.2000.01258.x
Differential regulation of nitric oxide synthase isoforms in experimental acute chagasic cardiomyopathy
Abstract
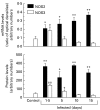
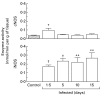
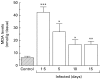
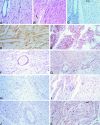
We have previously demonstrated induction and high level expression of IL-1beta, IL-6 and tumour necrosis factor-alpha in the myocardium during the acute stage of experimental Trypanosoma cruzi infection (Chagas' disease). The myocardial depressive effects of these cytokines are mediated in part by the induction of nitric oxide synthase (NOS), production of nitric oxide (NO) and formation of peroxynitrite. In this study we investigated the expression, activity and localization of NOS isoforms, and the levels of NO, malondialdehyde (a measure of oxidative stress), and peroxynitrite in rats at 1.5, 5, 10 and 15 days after infection with T. cruzi trypomastigotes. The myocardial inflammatory infiltrate and number of amastigote nests increased over the course of infection. A significant increase in tissue nitrate + nitrite levels, NOS2 mRNA, and NOS2 enzyme activity was observed at all time points in the infected compared with uninfected animals. The enzyme activity of constitutive NOS, tissue malondialdehyde levels, and NOS3 mRNA levels was only transiently increased after infection. The protein levels of the NOS isoforms paralleled their mRNA expression. While no positive nitrotyrosine immunoreactivity was detected in control myocardium, its levels increased in infected animals over time. Thus, by 1.5 days post-infection, when no parasite or immune cell infiltration could be detected, the myocardium expressed high levels of NOS and NO metabolites. Nevertheless, the early production of NO in the myocardium was not sufficient to clear the parasites.
Figures


Similar articles
-
Role of NO synthase in the development of Trypanosoma cruzi-induced cardiomyopathy in mice.Am J Trop Med Hyg. 2009 May;80(5):782-7. Am J Trop Med Hyg. 2009. PMID: 19407124 Free PMC article.
-
Temporal expression of pro-inflammatory cytokines and inducible nitric oxide synthase in experimental acute Chagasic cardiomyopathy.Am J Pathol. 1998 Apr;152(4):925-34. Am J Pathol. 1998. PMID: 9546353 Free PMC article.
-
Time course and cellular localization of inducible nitric oxide synthases expression during cardiac allograft rejection.Ann Thorac Surg. 1999 Mar;67(3):716-22. doi: 10.1016/s0003-4975(98)01346-0. Ann Thorac Surg. 1999. PMID: 10215216
-
Expressional control of the 'constitutive' isoforms of nitric oxide synthase (NOS I and NOS III).FASEB J. 1998 Jul;12(10):773-90. FASEB J. 1998. PMID: 9657518 Review.
-
Molecular biology of nitric oxide synthases.Cancer Metastasis Rev. 1998 Mar;17(1):7-23. doi: 10.1023/a:1005940202801. Cancer Metastasis Rev. 1998. PMID: 9544420 Review.
Cited by
-
Unveiling Lovastatin's Anti-Inflammatory Potential in Mouse's Brain during Acute Trypanosoma cruzi Infection.Biology (Basel). 2024 Apr 27;13(5):301. doi: 10.3390/biology13050301. Biology (Basel). 2024. PMID: 38785783 Free PMC article.
-
Evasion and Immuno-Endocrine Regulation in Parasite Infection: Two Sides of the Same Coin in Chagas Disease?Front Microbiol. 2016 May 23;7:704. doi: 10.3389/fmicb.2016.00704. eCollection 2016. Front Microbiol. 2016. PMID: 27242726 Free PMC article. Review.
-
Cellular Stress and Senescence Induction during Trypanosoma cruzi Infection.Trop Med Infect Dis. 2022 Jul 11;7(7):129. doi: 10.3390/tropicalmed7070129. Trop Med Infect Dis. 2022. PMID: 35878141 Free PMC article. Review.
-
Strategies of Pathogens to Escape from NO-Based Host Defense.Antioxidants (Basel). 2022 Nov 3;11(11):2176. doi: 10.3390/antiox11112176. Antioxidants (Basel). 2022. PMID: 36358549 Free PMC article. Review.
-
Role of NO synthase in the development of Trypanosoma cruzi-induced cardiomyopathy in mice.Am J Trop Med Hyg. 2009 May;80(5):782-7. Am J Trop Med Hyg. 2009. PMID: 19407124 Free PMC article.
References
-
- Andrade ZA. Pathogenesis of Chagas' disease. Res Immunol. 1991;142:126–9. - PubMed
-
- Hagar JM, Rahimtoola SH. Chagas heart disease. Curr Probl Cardiol. 1995;20:827–924. - PubMed
-
- Chandrasekar B, Melby PC, Troyer DA, Freeman GL. Induction of proinflammatory cytokine expression in experimental acute Chagasic cardiomyopathy. Biochem Biophys Res Commun. 1996;223:365–71. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

