In vivo formation of complex microvessels lined by human endothelial cells in an immunodeficient mouse
- PMID: 10890921
- PMCID: PMC16844
- DOI: 10.1073/pnas.150242297
In vivo formation of complex microvessels lined by human endothelial cells in an immunodeficient mouse
Abstract
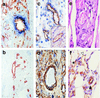
We have identified conditions for forming cultured human umbilical vein endothelial cells (HUVEC) into tubes within a three-dimensional gel that on implantation into immunoincompetent mice undergo remodeling into complex microvessels lined by human endothelium. HUVEC suspended in mixed collagen/fibronectin gels organize into cords with early lumena by 24 h and then apoptose. Twenty-hour constructs, s.c. implanted in immunodeficient mice, display HUVEC-lined thin-walled microvessels within the gel 31 days after implantation. Retroviral-mediated overexpression of a caspase-resistant Bcl-2 protein delays HUVEC apoptosis in vitro for over 7 days. Bcl-2-transduced HUVEC produce an increased density of HUVEC-lined perfused microvessels in vivo compared with untransduced or control-transduced HUVEC. Remarkably, Bcl-2- but not control-transduced HUVEC recruit an ingrowth of perivascular smooth-muscle alpha-actin-expressing mouse cells at 31 days, which organize by 60 days into HUVEC-lined multilayered structures resembling true microvessels. This system provides an in vivo model for dissecting mechanisms of microvascular remodeling by using genetically modified endothelium. Incorporation of such human endothelial-lined microvessels into engineered synthetic skin may improve graft viability, especially in recipients with impaired angiogenesis.
Figures



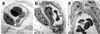
Comment in
-
Synthesis of organs: in vitro or in vivo?Proc Natl Acad Sci U S A. 2000 Aug 15;97(17):9354-6. doi: 10.1073/pnas.180313497. Proc Natl Acad Sci U S A. 2000. PMID: 10931961 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

