Translational control of C/EBPalpha and C/EBPbeta isoform expression
- PMID: 10921906
- PMCID: PMC316813
Translational control of C/EBPalpha and C/EBPbeta isoform expression
Abstract
Transcription factors derived from CCAAT/enhancer binding protein (C/EBP)alpha and C/EBPbeta genes control differentiation and proliferation in a number of cell types. Various C/EBP isoforms arise from unique C/EBPbeta and C/EBPalpha mRNAs by differential initiation of translation. These isoforms retain different parts of the amino terminus and therefore display different functions in gene regulation and proliferation control. We show that PKR and mTOR signaling pathways control the ratio of C/EBP isoform expression through the eukaryotic translation initiation factors eIF-2alpha and eIF-4E, respectively. An evolutionary conserved upstream open reading frame in C/EBPalpha and C/EBPbeta mRNAs is a prerequisite for regulated initiation from the different translation initiation sites and integrates translation factor activity. Deregulated translational control leading to aberrant C/EBPalpha and C/EBPbeta isoform expression or ectopic expression of truncated isoforms disrupts terminal differentiation and induces a transformed phenotype in 3T3-L1 cells. Our results demonstrate that the translational controlled ratio of C/EBPalpha and C/EBPbeta isoform expression determines cell fate.
Figures





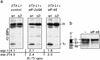
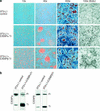

References
-
- Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K. Current Protocols in Molecular Biology. New York, NY: Green Publishing Associates/John Wiley & Sons; 1993.
-
- Baer M, Williams SC, Dillner A, Schwartz RC, Johnson PF. Autocrine signals control CCAAT/enhancer binding protein beta expression, localization, and activity in macrophages. Blood. 1998;92:4353–4365. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
