The tumor suppressor p53 can both stimulate and inhibit ultraviolet light-induced apoptosis
- PMID: 10930452
- PMCID: PMC14938
- DOI: 10.1091/mbc.11.8.2543
The tumor suppressor p53 can both stimulate and inhibit ultraviolet light-induced apoptosis
Abstract
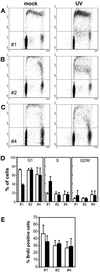
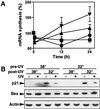
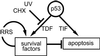
We have previously shown that the tumor suppressor p53 can play a protective role against UV-induced apoptosis in human fibroblasts. In the present study, we investigated whether the protective function of p53 expression is established before or after UV irradiation. Using a stable human cell line expressing a murine temperature-sensitive p53 in which p53 function could be tightly and reversibly regulated, we found that functional p53 stimulated the induction of apoptosis when expressed for as little as 4-12 h after UV irradiation and that this induction was not dependent on de novo protein synthesis. In contrast, expression of p53 for 12 h or more before UV irradiation reduced the extent of apoptosis even when functional p53 expression was maintained after irradiation. The protection conferred by p53 required ongoing protein synthesis and correlated with enhanced recovery of mRNA synthesis. Together, these results suggest that p53 induces distinct proapoptotic and antiapoptotic signals and that these opposing activities can be separated both temporally and by their requirement for de novo protein synthesis. These findings may have important implications for the refinement of gene therapy approaches combining p53 with pharmacological agents that target transcription or translation.
Figures




Similar articles
-
Role for p53 in the recovery of transcription and protection against apoptosis induced by ultraviolet light.Neoplasia. 1999 Aug;1(3):276-84. doi: 10.1038/sj.neo.7900028. Neoplasia. 1999. PMID: 10935482 Free PMC article.
-
Regulation of Bax activation and apoptotic response to UV irradiation by p53 transcription-dependent and -independent pathways.Cancer Lett. 2008 Nov 28;271(2):231-9. doi: 10.1016/j.canlet.2008.06.006. Epub 2008 Jul 25. Cancer Lett. 2008. PMID: 18657356
-
UV but not gamma irradiation accelerates p53-induced apoptosis of teratocarcinoma cells by repressing MDM2 transcription.Cancer Res. 2000 Nov 1;60(21):6184-8. Cancer Res. 2000. PMID: 11085543
-
P53 plays a protective role against UV- and cisplatin-induced apoptosis in transcription-coupled repair proficient fibroblasts.Oncogene. 2001 Oct 11;20(46):6805-8. doi: 10.1038/sj.onc.1204901. Oncogene. 2001. PMID: 11709715
-
p53-mediated protective responses to UV irradiation.Proc Natl Acad Sci U S A. 1997 Nov 11;94(23):12255-7. doi: 10.1073/pnas.94.23.12255. Proc Natl Acad Sci U S A. 1997. PMID: 9356435 Free PMC article. Review. No abstract available.
Cited by
-
The role of mRNA decay in p53-induced gene expression.RNA. 2011 Dec;17(12):2222-34. doi: 10.1261/rna.030122.111. Epub 2011 Oct 21. RNA. 2011. PMID: 22020975 Free PMC article.
-
UVB-mediated activation of p38 mitogen-activated protein kinase enhances resistance of normal human keratinocytes to apoptosis by stabilizing cytoplasmic p53.Biochem J. 2002 Jul 1;365(Pt 1):133-45. doi: 10.1042/BJ20020072. Biochem J. 2002. PMID: 12071847 Free PMC article.
-
A Temperature Sensitive Variant of p53 Drives p53-Dependent MicroRNA Expression without Evidence of Widespread Post-Transcriptional Gene Silencing.PLoS One. 2016 Feb 3;11(2):e0148529. doi: 10.1371/journal.pone.0148529. eCollection 2016. PLoS One. 2016. PMID: 26840126 Free PMC article.
-
DNA damage-induced ATM- and Rad-3-related (ATR) kinase activation in non-replicating cells is regulated by the XPB subunit of transcription factor IIH (TFIIH).J Biol Chem. 2017 Jul 28;292(30):12424-12435. doi: 10.1074/jbc.M117.788406. Epub 2017 Jun 7. J Biol Chem. 2017. PMID: 28592488 Free PMC article.
-
p53 is a chromatin accessibility factor for nucleotide excision repair of DNA damage.EMBO J. 2003 Feb 17;22(4):975-86. doi: 10.1093/emboj/cdg082. EMBO J. 2003. PMID: 12574133 Free PMC article.
References
-
- Aranda-Anzaldo A, Orozco-Velasco F, Garcia-Villa E, Gariglio P. p53 is a rate-limiting factor in the repair of higher-order DNA structure. Biochim Biophys Acta. 1999;1446:181–192. - PubMed
-
- Barley RDC, Enns L, Paterson MC, Mirzayans R. Aberrant p21(WAF1)-dependent growth arrest as the possible mechanism of abnormal resistance to ultraviolet light cytotoxicity in Li-Fraumeni syndrome fibroblast strains heterozygous for TP53 mutations. Oncogene. 1998;17:533–543. - PubMed
-
- Bissonnette N, Hunting DJ. p21-induced cycle arrest in G1 protects cells from apoptosis induced by UV-irradiation or RNA polymerase II blockage. Oncogene. 1998;16:3461–3469. - PubMed
-
- Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP, Sedivy JM, Kinzler KW, Vogelstein B. Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science. 1998;282:1497–1501. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

