Pharmacokinetics of epimeric budesonide and fluticasone propionate after repeat dose inhalation--intersubject variability in systemic absorption from the lung
- PMID: 10930963
- PMCID: PMC2014401
- DOI: 10.1046/j.1365-2125.2000.00218.x
Pharmacokinetics of epimeric budesonide and fluticasone propionate after repeat dose inhalation--intersubject variability in systemic absorption from the lung
Abstract
Aims: Pharmacokinetic variability is likely to be a significant factor contributing to the interindividual differences in dose requirements, anti-inflammatory response and side-effects with inhaled corticosteroids (ICS), but there is limited information about the disposition of ICS during regular dosing with a pressurized metered dose inhaler (pMDI). This study uses a mixed effects modelling approach to quantify and compare the interindividual variability in pharmacokinetics of epimeric budesonide (BUD) and fluticasone propionate (FP) after repeat-dose inhalation.
Methods: This pharmacokinetic substudy was part of a previously published open-label, randomised, placebo-controlled, 7-period crossover study to evaluate the short-term effects on plasma cortisol levels of inhaled BUD (400, 800, 1600 microg twice daily) and FP (375, 750, 1000 microg twice daily) via pMDI in a group of healthy male volunteers. On the fifth day of each high-dose treatment period (BUD 1600 microg twice daily and FP 1000 microg twice daily), venous blood samples were collected in nine subjects prior to the last dose and at 15 min, 30 min, 1, 2, 4, 6 and 8 h postdose for measurement of plasma drug concentrations to determine the pharmacokinetics of epimeric BUD and FP following inhalation. Non-compartmental analysis and a mixed effects model were used to characterize the disposition profiles.
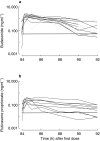
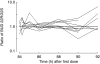
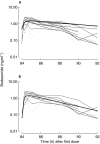
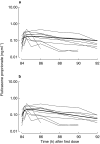
Results: Both drugs had a rapid absorption half-life (BUD 10 min vs FP 11.3 min), but quite different elimination half-lives (BUD 2.4 h vs FP 7.8 h). Although there were intraindividual differences in the handling of the 22R-and 22S-epimers of BUD, there were no consistent pharmacokinetic differences between the two enantiomers in the group as a whole. Consistent with previous reports of FP's higher volume of distribution (V) and lower systemic bioavailability (F), the V/F ratio was lower for BUD than FP (498 l vs 8100 l). The parameter with the greatest interindividual variability for both BUD and FP was the rate of systemic absorption from the lung.
Conclusions: This is the first report describing the pharmacokinetics of epimeric BUD and FP after repeat dose inhalation via pMDI. Three observations may be of clinical relevance: (1) there is considerable intersubject variability in the rate of absorption of both drugs from the lung; (2) in some individuals there was a long t(1/2),z for BUD, resulting in higher and more sustained plasma drug levels in the 4-12 h postdose period than would be predicted from single-dose pharmacokinetic data; and (3) there is evidence of diurnal variation in FP pharmacokinetics, with higher-than-expected plasma drug concentrations in the morning compared with the evening.
Figures
Similar articles
-
Single-dose and steady-state pharmacokinetic and pharmacodynamic evaluation of therapeutically clinically equivalent doses of inhaled fluticasone propionate and budesonide, given as Diskus or Turbohaler dry-powder inhalers to healthy subjects.J Clin Pharmacol. 2001 Dec;41(12):1329-38. doi: 10.1177/00912700122012913. J Clin Pharmacol. 2001. PMID: 11762560 Clinical Trial.
-
Pharmacokinetics and systemic activity of fluticasone via Diskus and pMDI, and of budesonide via Turbuhaler.Br J Clin Pharmacol. 2001 Nov;52(5):529-38. doi: 10.1046/j.0306-5251.2001.01493.x. Br J Clin Pharmacol. 2001. PMID: 11736861 Free PMC article. Clinical Trial.
-
Lung deposition and systemic availability of fluticasone Diskus and budesonide Turbuhaler in children.Am J Respir Crit Care Med. 2003 Oct 1;168(7):779-82. doi: 10.1164/rccm.200302-200OC. Epub 2003 Jul 31. Am J Respir Crit Care Med. 2003. PMID: 12893646 Clinical Trial.
-
Clinical pharmacokinetics of inhaled budesonide.Clin Pharmacokinet. 2001;40(6):427-40. doi: 10.2165/00003088-200140060-00004. Clin Pharmacokinet. 2001. PMID: 11475468 Review.
-
Inhaled salmeterol/fluticasone propionate combination: a review of its use in persistent asthma.Drugs. 2000 Nov;60(5):1207-33. doi: 10.2165/00003495-200060050-00012. Drugs. 2000. PMID: 11129128 Review.
Cited by
-
How does race and ethnicity effect the precision treatment of asthma?Expert Rev Precis Med Drug Dev. 2019;4(6):337-356. doi: 10.1080/23808993.2019.1690396. Epub 2019 Nov 14. Expert Rev Precis Med Drug Dev. 2019. PMID: 33015363 Free PMC article.
-
Pharmacokinetics of [14C]ciclesonide after oral and intravenous administration to healthy subjects.Clin Pharmacokinet. 2004;43(7):479-86. doi: 10.2165/00003088-200443070-00004. Clin Pharmacokinet. 2004. PMID: 15139796 Clinical Trial.
-
Corticosteroids inhibit anti-IgE activities of specialized proresolving mediators on B cells from asthma patients.JCI Insight. 2017 Feb 9;2(3):e88588. doi: 10.1172/jci.insight.88588. JCI Insight. 2017. PMID: 28194434 Free PMC article.
-
Assessment of the predictive capability of modelling and simulation to determine bioequivalence of inhaled drugs: A systematic review.Daru. 2022 Jun;30(1):229-243. doi: 10.1007/s40199-021-00423-7. Epub 2022 Jan 30. Daru. 2022. PMID: 35094370 Free PMC article.
-
Towards a Quantitative Mechanistic Understanding of Localized Pulmonary Tissue Retention-A Combined In Vivo/In Silico Approach Based on Four Model Drugs.Pharmaceutics. 2020 Apr 29;12(5):408. doi: 10.3390/pharmaceutics12050408. Pharmaceutics. 2020. PMID: 32365674 Free PMC article.
References
-
- Edsbacker S, Andersson KE, Ryrfeldt A. Nasal bioavailability and systemic effects of the glucocorticoid budesonide in man. Eur J Clin Pharmacol. 1995;29:477–481. - PubMed
-
- Chisholm D, Chalkley S. Cushing’s syndrome from an inhaled glucocorticoid. Med J Aust. 1994;161:232. - PubMed
-
- Donnelly R, Williams KM, Baker AB, Badcock CA, Day RO, Seale JP. Effects of budesonide and fluticasone on 24-hour plasma cortisol. A dose–response study. Am J Resp Crit Care Med. 1997;156:1746–1751. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

