Functional profile of the giant metacerebral neuron of Helix aspersa: temporal and spatial dynamics of electrical activity in situ
- PMID: 10944170
- PMCID: PMC2270048
- DOI: 10.1111/j.1469-7793.2000.00055.x
Functional profile of the giant metacerebral neuron of Helix aspersa: temporal and spatial dynamics of electrical activity in situ
Abstract
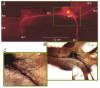
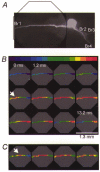
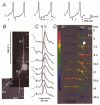
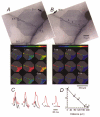
1. Understanding the biophysical properties of single neurons and how they process information is fundamental to understanding how the brain works. However, action potential initiation and the preceding integration of the synaptic signals in neuronal processes of individual cells are complex and difficult to understand in the absence of detailed, spatially resolved measurements. Multi-site optical recording with voltage-sensitive dyes from individual neurons in situ was used to provide these kinds of measurements. We analysed in detail the pattern of initiation and propagation of spikes evoked synaptically in an identified snail (Helix aspersa) neuron in situ. 2. Two main spike trigger zones were identified. The trigger zones were activated selectively by different sets of synaptic inputs which also produced different spike propagation patterns. 3. Synaptically evoked action potentials did not always invade all parts of the neuron. The conduction of the axonal spike was regularly blocked at particular locations on neuronal processes. 4. The propagating spikes in some axonal branches consistently reversed direction at certain branch points, a phenomenon known as reflection. 5. These experimental results, when linked to a computer model, could allow a new level of analysis of the electrical structure of single neurons.
Figures



References
-
- Altrup U, Peters M. Procedure of intracellular staining of neurons in the snail Helix pomatia. Journal of Neuroscience Methods. 1982;5:161–165. - PubMed
-
- Baccus SA, Burrell BD, Sahley CL, Muller KJ. Action potential reflection and failure at axon branch points cause stepwise changes in EPSPs in a neuron essential for learning. Journal of Neurophysiology. 2000;83:1693–1700. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

