Endogenously produced nitric oxide inhibits endothelial cell growth as demonstrated using novel antisense cell lines
- PMID: 10960079
- PMCID: PMC1572298
- DOI: 10.1038/sj.bjp.0703539
Endogenously produced nitric oxide inhibits endothelial cell growth as demonstrated using novel antisense cell lines
Abstract
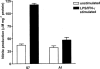
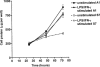
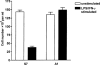
Proliferation of endothelial cells is a vital component of vascular repair and angiogenesis. The endothelial cell mediator, nitric oxide (NO) has been reported both to inhibit and to promote endothelial cell proliferation. In this study we have generated cell lines which constitutively express antisense RNA to a region of inducible nitric oxide synthase (iNOS) from a murine endothelial cell line, sEnd-1. In response to stimulation with lipopolysaccharide (LPS) and interferon-gamma (IFN-gamma) these antisense cells had no detectable RNA for endogenous iNOS, barely detectable iNOS protein and produced 82% less NO than did the control transfected line. Stimulation of the control transfected line caused significant NO production and inhibition of cell growth whereas for the antisense line, producing little NO in response to stimulation, proliferation remained the same as for unstimulated cells. No differences in cell death were observed between unstimulated and LPS/IFN-gamma stimulated cells. The data presented in this study directly demonstrate that NO derived endogenously from iNOS inhibits proliferation of endothelial cells. This approach overcomes problems in other studies where NO donors or non-isoform specific inhibitors of NO synthase have been used.
Figures


References
-
- BOGLE R.G., MACALLISTER R.J., WHITLEY G.ST.J., VALLANCE P. Induction of NG-monomethyl-L-arginine uptake: a mechanism of differential inhibition of NO synthases. Am. J. Physiol. 1995;269:C750–C756. - PubMed
-
- BRADFORD M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilising the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. - PubMed
-
- COOKE J.P., DZAU V.J. Nitric oxide synthase: role in genesis of vascular disease. Ann. Rev. Med. 1997;48:489–509. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

