Severed channels probe regulation of gating of cystic fibrosis transmembrane conductance regulator by its cytoplasmic domains
- PMID: 10962022
- PMCID: PMC2233695
- DOI: 10.1085/jgp.116.3.477
Severed channels probe regulation of gating of cystic fibrosis transmembrane conductance regulator by its cytoplasmic domains
Abstract
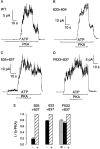
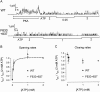
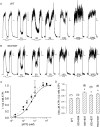
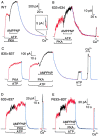
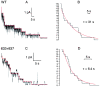
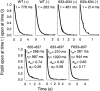
Opening and closing of a CFTR Cl(-) channel is controlled by PKA-mediated phosphorylation of its cytoplasmic regulatory (R) domain and by ATP binding, and likely hydrolysis, at its two nucleotide binding domains. Functional interactions between the R domain and the two nucleotide binding domains were probed by characterizing the gating of severed CFTR channels expressed in Xenopus oocytes. Expression levels were assessed using measurements of oocyte conductance, and detailed functional characteristics of the channels were extracted from kinetic analyses of macroscopic current relaxations and of single-channel gating events in membrane patches excised from the oocytes. The kinetic behavior of wild-type (WT) CFTR channels was compared with that of split CFTR channels bearing a single cut (between residues 633 and 634) just before the R domain, of split channels with a single cut (between residues 835 and 837) just after the R domain, and of split channels from which the entire R domain (residues 634-836) between those two cut sites was omitted. The channels cut before the R domain had characteristics almost identical to those of WT channels, except for less than twofold shorter open burst durations in the presence of PKA. Channels cut just after the R domain were characterized by a low level of activity even without phosphorylation, strong stimulation by PKA, enhanced apparent affinity for ATP as assayed by open probability, and a somewhat destabilized binding site for the locking action of the nonhydrolyzable ATP analog AMPPNP. Split channels with no R domain (from coexpression of CFTR segments 1-633 and 837-1480) were highly active without phosphorylation, but otherwise displayed the characteristics of channels cut after the R domain, including higher apparent ATP affinity, and less tight binding of AMPPNP at the locking site, than for WT. Intriguingly, severed channels with no R domain were still noticeably stimulated by PKA, implying that activation of WT CFTR by PKA likely also includes some component unrelated to the R domain. As the maximal opening rates were the same for WT channels and split channels with no R domain, it seems that the phosphorylated R domain does not stimulate opening of CFTR channels; rather, the dephosphorylated R domain inhibits them.
Figures
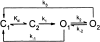
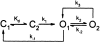





Similar articles
-
Functional roles of nonconserved structural segments in CFTR's NH2-terminal nucleotide binding domain.J Gen Physiol. 2005 Jan;125(1):43-55. doi: 10.1085/jgp.200409174. Epub 2004 Dec 13. J Gen Physiol. 2005. PMID: 15596536 Free PMC article.
-
Preferential phosphorylation of R-domain Serine 768 dampens activation of CFTR channels by PKA.J Gen Physiol. 2005 Feb;125(2):171-86. doi: 10.1085/jgp.200409076. Epub 2005 Jan 18. J Gen Physiol. 2005. PMID: 15657296 Free PMC article.
-
A cluster of negative charges at the amino terminal tail of CFTR regulates ATP-dependent channel gating.J Physiol. 2001 Oct 15;536(Pt 2):459-70. doi: 10.1111/j.1469-7793.2001.0459c.xd. J Physiol. 2001. PMID: 11600681 Free PMC article.
-
ATP hydrolysis cycles and the gating of CFTR Cl- channels.Acta Physiol Scand Suppl. 1998 Aug;643:247-56. Acta Physiol Scand Suppl. 1998. PMID: 9789567 Review.
-
The gating of the CFTR channel.Cell Mol Life Sci. 2017 Jan;74(1):85-92. doi: 10.1007/s00018-016-2390-z. Epub 2016 Oct 1. Cell Mol Life Sci. 2017. PMID: 27696113 Free PMC article. Review.
Cited by
-
CFTR (ABCC7) is a hydrolyzable-ligand-gated channel.Pflugers Arch. 2007 Feb;453(5):693-702. doi: 10.1007/s00424-006-0140-z. Epub 2006 Sep 26. Pflugers Arch. 2007. PMID: 17021796 Review.
-
Molecular structure of the ATP-bound, phosphorylated human CFTR.Proc Natl Acad Sci U S A. 2018 Dec 11;115(50):12757-12762. doi: 10.1073/pnas.1815287115. Epub 2018 Nov 20. Proc Natl Acad Sci U S A. 2018. PMID: 30459277 Free PMC article.
-
Phosphorylation of CFTR by PKA promotes binding of the regulatory domain.EMBO J. 2005 Aug 3;24(15):2730-40. doi: 10.1038/sj.emboj.7600747. Epub 2005 Jul 7. EMBO J. 2005. PMID: 16001079 Free PMC article.
-
Cysteine accessibility probes timing and extent of NBD separation along the dimer interface in gating CFTR channels.J Gen Physiol. 2015 Apr;145(4):261-83. doi: 10.1085/jgp.201411347. J Gen Physiol. 2015. PMID: 25825169 Free PMC article.
-
The allosteric regulatory mechanism of the Escherichia coli MetNI methionine ATP binding cassette (ABC) transporter.J Biol Chem. 2015 Apr 3;290(14):9135-40. doi: 10.1074/jbc.M114.603365. Epub 2015 Feb 12. J Biol Chem. 2015. PMID: 25678706 Free PMC article.
References
-
- Aleksandrov A.A., Riordan J.R. Regulation of CFTR ion channel gating by MgATP. FEBS Lett. 1998;431:97–101. - PubMed
-
- Ball F.G., Sansom M.S.P. Ion-channel gating mechanismsmodel identification and parameter estimation from single channel recordings. Proc. R. Soc. Lond. B Biol. Sci. 1989;236:385–416. - PubMed
-
- Carson M.R., Welsh M.J. 5′-adenylylimidodiphosphate does not activate CFTR chloride channels in cell-free patches of membrane. Am. J. Physiol. Lung Cell. Mol. Physiol. 1993;265:L27–L32. - PubMed
-
- Carson M.R., Travis S.M., Welsh M.J. The two nucleotide-binding domains of cystic fibrosis transmembrane conductance regulator (CFTR) have distinct functions in controlling channel activity. J. Biol. Chem. 1995;270:1711–1717. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

