Cotranslational dimerization of the Rel homology domain of NF-kappaB1 generates p50-p105 heterodimers and is required for effective p50 production
- PMID: 10970863
- PMCID: PMC302078
- DOI: 10.1093/emboj/19.17.4712
Cotranslational dimerization of the Rel homology domain of NF-kappaB1 generates p50-p105 heterodimers and is required for effective p50 production
Abstract
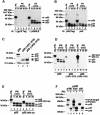
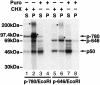
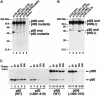
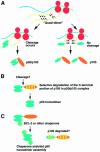
Generation of the NF-kappaB p50 transcription factor is mediated by the proteasome. We found previously that p50 is generated during translation of the NFKB1 gene and that this cotranslational processing allows the production of both p50 and p105 from a single mRNA. We now demonstrate that the Rel homology domain in p50 undergoes cotranslational dimerization and that this interaction is required for efficient production of p50. We further show that this coupling of dimerization and proteasome processing during translation uniquely generates p50-p105 heterodimers. Accordingly, after the primary cotranslational event, additional posttranslational steps regulate p50 homodimer formation and the intracellular ratio of p50 and p105. This cellular strategy places p50 under the control of the p105 inhibitor early in its biogenesis, thereby regulating the pool of p50 homodimers within the cell.
Figures




References
-
- Baeuerle P.A. and Baltimore,D. (1988) IκB: a specific inhibitor of the NF-κB transcription factor. Science, 242, 540–546. - PubMed
-
- Baeuerle P.A. and Baltimore,D. (1989) A 65-kD subunit of active NF-κB is required for inhibition of NF–κB by IκB. Genes Dev., 3, 1689–1698. - PubMed
-
- Baumeister W., Walz,J., Zühl,F. and Seemüller,E. (1998) The proteasome: paradigm of a self-compartmentalizing protease. Cell, 92, 367–380. - PubMed
-
- Belich M.P., Salmerón,A., Johnson,L.H. and Ley,S.C. (1999) TPL-2 kinase regulates the proteolysis of the NF-κB-inhibitory protein NF-κB 1 p105. Nature, 397, 363–368. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

