Islet xenograft destruction in the hu-PBL-severe combined immunodeficient (SCID) mouse necessitates anti-CD3 preactivation of human immune cells
- PMID: 10971525
- PMCID: PMC1905726
- DOI: 10.1046/j.1365-2249.2000.01300.x
Islet xenograft destruction in the hu-PBL-severe combined immunodeficient (SCID) mouse necessitates anti-CD3 preactivation of human immune cells
Abstract
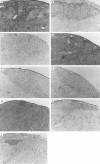
Introduction of the hu-PBL-SCID mouse model has yielded a potentially useful tool for research in transplantation. The aim of this study was to define the conditions necessary for a reconstituted human immune system to destroy in a consistent manner rat islet xenografts in the alloxan-diabetic hu-PBL-SCID mouse. We examined different time points of hu-PBL reconstitution, different transplantation sites of the islets and several hu-PBL reconstitution protocols. Major differences in graft destruction were observed between the different hu-PBL reconstitution protocols, irrespective of timing of hu-PBL reconstitution or site of transplantation. Although preactivation of hu-PBL did not improve the level of hu-PBL chimerism, histological and immunohistochemical analysis of the grafts revealed a severe human lymphocytic infiltration and beta cell destruction only in the grafts of mice receiving preactivated hu-PBL. This beta cell injury resulted in impaired glucose tolerance, with in some animals recurrence of hyperglycaemia, and decreased insulin and C-peptide levels after glucose stimulation. Therefore, we conclude that activation of hu-PBL prior to transfer is essential in achieving xenograft infiltration and destruction in hu-PBL-SCID mice. The need for immune manipulation suggests that interactions between hu-PBL and xenografts in this model may be hampered by incompatibilities in cross-species adhesion and/or activation signals.
Figures
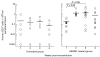



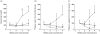
Similar articles
-
Human peripheral blood leukocyte-reconstituted severe combined immunodeficient mouse: analysis of the human immune response against porcine islet transplantation.Transplantation. 1997 Mar 27;63(6):818-23. doi: 10.1097/00007890-199703270-00005. Transplantation. 1997. PMID: 9089220
-
Human peripheral blood lymphocyte reconstituted severe combined immunodeficient (hu-PBL-SCID) mice. A model for human islet allograft rejection.Transplantation. 1994 Jun 15;57(11):1555-62. Transplantation. 1994. PMID: 8009588
-
Human peripheral blood lymphocyte-reconstituted, severe combined immunodeficient mice as a model for porcine islet xenograft rejection in humans.Artif Organs. 1996 Aug;20(8):878-82. doi: 10.1111/j.1525-1594.1996.tb04562.x. Artif Organs. 1996. PMID: 8853799
-
Xenogeneic PBL-scid mice: their potential and current limitations.Lab Anim Sci. 1993 Apr;43(2):151-5. Lab Anim Sci. 1993. PMID: 8320963 Review.
-
The SCID-hu mouse: an in vivo model for HIV-1 infection in humans.Mol Med Today. 1997 Jun;3(6):246-53. doi: 10.1016/s1357-4310(97)01046-0. Mol Med Today. 1997. PMID: 9211415 Review.
Cited by
-
Vitamin D deficiency in early life accelerates Type 1 diabetes in non-obese diabetic mice.Diabetologia. 2004 Mar;47(3):451-462. doi: 10.1007/s00125-004-1329-3. Epub 2004 Jan 31. Diabetologia. 2004. PMID: 14758446
-
Treatment of autoimmune diabetes recurrence in non-obese diabetic mice by mouse interferon-beta in combination with an analogue of 1alpha,25-dihydroxyvitamin-D3.Clin Exp Immunol. 2002 May;128(2):213-20. doi: 10.1046/j.1365-2249.2002.01825.x. Clin Exp Immunol. 2002. PMID: 11985511 Free PMC article.
-
Four-color flow cytometric analysis of peripheral blood donor cell chimerism.Hum Immunol. 2003 Aug;64(8):787-95. doi: 10.1016/s0198-8859(03)00113-7. Hum Immunol. 2003. PMID: 12878357 Free PMC article.
-
Progress, implications, and challenges in using humanized immune system mice in CAR-T therapy-Application evaluation and improvement.Animal Model Exp Med. 2024 Feb;7(1):3-11. doi: 10.1002/ame2.12353. Epub 2023 Oct 12. Animal Model Exp Med. 2024. PMID: 37823214 Free PMC article. Review.
-
Evaluation of the tolerogenic effects of donor bone marrow cells using a severe combined immunodeficient mouse-human islet transplant model.Hum Immunol. 2008 Oct;69(10):605-13. doi: 10.1016/j.humimm.2008.07.003. Epub 2008 Aug 12. Hum Immunol. 2008. PMID: 18703102 Free PMC article.
References
-
- Casteels K, Waer M, Laureys J, et al. Prevention of autoimmune destruction of syngeneic islet grafts in spontaneously diabetic nonobese diabetic mice by a combination of a vitamin D3 analogue and cyclosporin. Transplantation. 1998;65:1225–32. - PubMed
-
- Salmela K. Pancreatic islet transplantation. Ann Chir Gynaecol. 1997;86:149–51. - PubMed
-
- Bosma GC, Custer RP, Bosma MJ. A severe combined immunodeficiency mutation in the mouse. Nature. 1983;301:527–30. - PubMed
-
- McCune JM, Namikawa R, Kaneshima H, Shultz LD, Lieberman M, Weissman IL. The SCID-hu mouse: murine model for the analysis of human hematolymphoid differentiation and function. Science. 1988;241:1632–9. - PubMed
-
- Mosier DE, Gulizia RJ, Baird SM, Wilson DB. Transfer of a functional human immune system to mice with severe combined immunodeficiency. Nature. 1988;335:256–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

