Two cell adhesion molecules, nectin and cadherin, interact through their cytoplasmic domain-associated proteins
- PMID: 10974003
- PMCID: PMC2175253
- DOI: 10.1083/jcb.150.5.1161
Two cell adhesion molecules, nectin and cadherin, interact through their cytoplasmic domain-associated proteins
Abstract
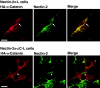
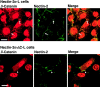
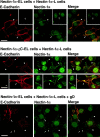
We have found a new cell-cell adhesion system at cadherin-based cell-cell adherens junctions (AJs) consisting of at least nectin and l-afadin. Nectin is a Ca(2+)-independent homophilic immunoglobulin-like adhesion molecule, and l-afadin is an actin filament-binding protein that connects the cytoplasmic region of nectin to the actin cytoskeleton. Both the trans-interaction of nectin and the interaction of nectin with l-afadin are necessary for their colocalization with E-cadherin and catenins at AJs. Here, we examined the mechanism of interaction between these two cell-cell adhesion systems at AJs by the use of alpha-catenin-deficient F9 cell lines and cadherin-deficient L cell lines stably expressing their various components. We showed here that nectin and E-cadherin were colocalized through l-afadin and the COOH-terminal half of alpha-catenin at AJs. Nectin trans-interacted independently of E-cadherin, and the complex of E-cadherin and alpha- and beta-catenins was recruited to nectin-based cell-cell adhesion sites through l-afadin without the trans-interaction of E-cadherin. Our results indicate that nectin and cadherin interact through their cytoplasmic domain-associated proteins and suggest that these two cell-cell adhesion systems cooperatively organize cell-cell AJs.
Figures


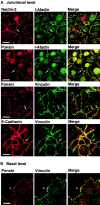
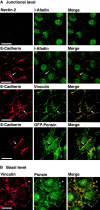
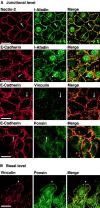

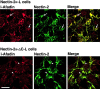
References
-
- Adra C.N., Boer P.H., McBurney M.W. Cloning and expression of the mouse pgk-1 gene and the nucleotide sequence of its promoter. Gene. 1987;60:65–74. - PubMed
-
- Aoki J., Koike S., Ise I., Sato-Yoshida Y., Nomoto A. Amino acid residues on human poliovirus receptor involved in interaction with poliovirus. J. Biol. Chem. 1994;269:8431–8438. - PubMed
-
- Aoki J., Koike S., Asou H., Ise I., Suwa H., Tanaka T., Miyasaka M., Nomoto A. Mouse homolog of poliovirus receptor-related gene 2 product, mPRR2, mediates homophilic cell aggregation. Exp. Cell Res. 1997;235:374–384. - PubMed
-
- Asakura T., Nakanishi H., Sakisaka T., Takahashi K., Mandai K., Nishimura M., Sasaki T., Takai Y. Similar and differential behavior between the nectin-afadin-ponsin and cadherin-catenin systems during the formation and disruption of the polarized junctional alignment in epithelial cells. Genes Cells. 1999;4:573–581. - PubMed
-
- Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

