Lymphocyte CC chemokine receptor 9 and epithelial thymus-expressed chemokine (TECK) expression distinguish the small intestinal immune compartment: Epithelial expression of tissue-specific chemokines as an organizing principle in regional immunity
- PMID: 10974041
- PMCID: PMC2193265
- DOI: 10.1084/jem.192.5.761
Lymphocyte CC chemokine receptor 9 and epithelial thymus-expressed chemokine (TECK) expression distinguish the small intestinal immune compartment: Epithelial expression of tissue-specific chemokines as an organizing principle in regional immunity
Abstract
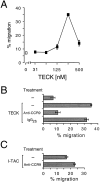
The immune system has evolved specialized cellular and molecular mechanisms for targeting and regulating immune responses at epithelial surfaces. Here we show that small intestinal intraepithelial lymphocytes and lamina propria lymphocytes migrate to thymus-expressed chemokine (TECK). This attraction is mediated by CC chemokine receptor (CCR)9, a chemoattractant receptor expressed at high levels by essentially all CD4(+) and CD8(+) T lymphocytes in the small intestine. Only a small subset of lymphocytes in the colon are CCR9(+), and lymphocytes from other tissues including tonsils, lung, inflamed liver, normal or inflamed skin, inflamed synovium and synovial fluid, breast milk, and seminal fluid are universally CCR9(-). TECK expression is also restricted to the small intestine: immunohistochemistry reveals that intense anti-TECK reactivity characterizes crypt epithelium in the jejunum and ileum, but not in other epithelia of the digestive tract (including stomach and colon), skin, lung, or salivary gland. These results imply a restricted role for lymphocyte CCR9 and its ligand TECK in the small intestine, and provide the first evidence for distinctive mechanisms of lymphocyte recruitment that may permit functional specialization of immune responses in different segments of the gastrointestinal tract. Selective expression of chemokines by differentiated epithelium may represent an important mechanism for targeting and specialization of immune responses.
Figures



References
-
- Zlotnik A., Yoshie O. Chemokinesa new classification system and their role in immunity. Immunity. 2000;12:121–127. - PubMed
-
- Campbell J.J., Butcher E.C. Chemokines in tissue-specific and microenvironment-specific lymphocyte homing. Curr. Opin. Immunol. 2000;12:336–341. - PubMed
-
- Campbell J.J., Haraldsen G., Pan J., Rottman J., Qin S., Ponath P.D., Andrew D.P., Warnke R., Ruffing N., Kassam N. The chemokine receptor CCR4 in vascular recognition by cutaneous but not intestinal memory T cells. Nature. 1999;400:776–780. - PubMed
-
- Zabel B.A., Agace W.W., Campbell J.J., Heath H.M., Parent D., Roberts A.I., Ebert E.C., Kassam N., Qin S., Zovko M. Human G protein–coupled receptor GPR-9-6/CC chemokine receptor 9 is selectively expressed on intestinal homing T lymphocytes, mucosal lymphocytes, and thymocytes and is required for thymus-expressed chemokine–mediated chemotaxis. J. Exp. Med. 1999;190:1241–1255. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

