Dynamics of transitional endoplasmic reticulum sites in vertebrate cells
- PMID: 10982397
- PMCID: PMC14972
- DOI: 10.1091/mbc.11.9.3013
Dynamics of transitional endoplasmic reticulum sites in vertebrate cells
Abstract
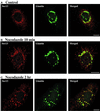
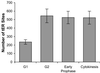
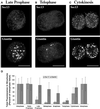
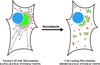
A typical vertebrate cell contains several hundred sites of transitional ER (tER). Presumably, tER sites generate elements of the ER-Golgi intermediate compartment (ERGIC), and ERGIC elements then generate Golgi cisternae. Therefore, characterizing the mechanisms that influence tER distribution may shed light on the dynamic behavior of the Golgi. We explored the properties of tER sites using Sec13 as a marker protein. Fluorescence microscopy confirmed that tER sites are long-lived ER subdomains. tER sites proliferate during interphase but lose Sec13 during mitosis. Unlike ERGIC elements, tER sites move very little. Nevertheless, when microtubules are depolymerized with nocodazole, tER sites redistribute rapidly to form clusters next to Golgi structures. Hence, tER sites have the unusual property of being immobile, yet dynamic. These findings can be explained by a model in which new tER sites are created by retrograde membrane traffic from the Golgi. We propose that the tER-Golgi system is organized by mutual feedback between these two compartments.
Figures




References
-
- Acharya U, Mallabiabarrena A, Acharya JK, Malhotra V. Signaling via mitogen-activated protein kinase kinase (MEK1) is required for Golgi fragmentation during mitosis. Cell. 1998;92:183–192. - PubMed
-
- Allan VJ, Schroer TA. Membrane motors. Curr Opin Cell Biol. 1999;11:476–482. - PubMed
-
- Andersen B, Osborn M, Weber K. Specific visualization of the distribution of the calcium dependent regulatory protein of cyclic nucleotide phosphodiesterase (modulator protein) in tissue culture cells by immuofluorescence microscopy: mitosis and intercellular bridge. Cytobiologie. 1978;17:354–364. - PubMed
-
- Balch WE, McCaffery JM, Plutner H, Farquhar MG. Vesicular stomatitis virus glycoprotein is sorted and concentrated during export from the endoplasmic reticulum. Cell. 1994;76:841–852. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

