Modulation of Akt kinase activity by binding to Hsp90
- PMID: 10995457
- PMCID: PMC27109
- DOI: 10.1073/pnas.170276797
Modulation of Akt kinase activity by binding to Hsp90
Abstract
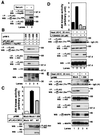
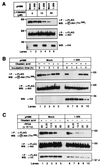
Serine/threonine kinase Akt/PKB is a downstream effector molecule of phosphoinositide 3-kinase and is thought to mediate many biological actions toward anti-apoptotic responses. We found that Akt formed a complex with a 90-kDa heat-shock protein (Hsp90) in vivo. By constructing deletion mutants, we identified that amino acid residues 229-309 of Akt were involved in the binding to Hsp90 and amino acid residues 327-340 of Hsp90beta were involved in the binding to Akt. Inhibition of Akt-Hsp90 binding led to the dephosphorylation and inactivation of Akt, which increased sensitivity of the cells to apoptosis-inducing stimulus. The dephosphorylation of Akt was caused by an increase in protein phosphatase 2A (PP2A)-mediated dephosphorylation and not by a decrease in 3-phosphoinositide-dependent protein kinase-1-mediated phosphorylation. These results indicate that Hsp90 plays an important role in maintaining Akt kinase activity by preventing PP2A-mediated dephosphorylation.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

