Role of perforin in controlling B-cell hyperactivity and humoral autoimmunity
- PMID: 10995792
- PMCID: PMC381389
- DOI: 10.1172/JCI8876
Role of perforin in controlling B-cell hyperactivity and humoral autoimmunity
Abstract
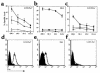
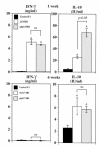
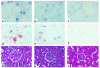
To determine the role of perforin-mediated cytotoxic T lymphocyte (CTL) effector function in immune regulation, we studied a well-characterized mouse model of graft-versus-host disease (GVHD). Induction of acute GVHD using perforin-deficient donor T cells (pfp-->F1) initially resulted in features of acute GVHD, e.g., engraftment of both donor CD4(+) and CD8(+) T cells, upregulation of Fas and FasL, production of antihost CTL, and secretion of both Th1 and Th2 cytokines. Despite fully functional FasL activity, pfp donor cells failed to totally eliminate host B cells, and, by 4 weeks of disease, cytokine production in pfp-->F1 mice had polarized to a Th2 response. Pfp-->F1 mice eventually developed features of chronic GVHD, such as increased numbers of B cells, persistence of donor CD4 T cells, autoantibody production, and lupuslike renal disease. We conclude that in the setting of B- and T-cell activation, perforin plays an important immunoregulatory role in the prevention of humoral autoimmunity through the elimination of both autoreactive B cells and ag-specific T cells. Moreover, an ineffective initial CTL response can evolve into a persistent antibody-mediated response and, with it, the potential for sustained humoral autoimmunity.
Figures

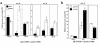
Comment in
-
Revisiting graft-versus-host disease models of autoimmunity: new insights in immune regulatory processes.J Clin Invest. 2000 Sep;106(6):745-7. doi: 10.1172/JCI11088. J Clin Invest. 2000. PMID: 10995784 Free PMC article. No abstract available.
References
-
- Millard PJ, Henkart MP, Reynolds CW, Henkart PA. Purification and properties of cytoplasmic granules from cytotoxic rat LGL tumors. J Immunol. 1984;132:3197–3204. - PubMed
-
- Clark WR, et al. Molecular pathways of CTL-mediated cytotoxicity. Immunol Rev. 1995;146:33–44. - PubMed
-
- Kagi D, et al. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin-deficient mice. Nature. 1994;369:31–37. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

