Reovirus-induced G(2)/M cell cycle arrest requires sigma1s and occurs in the absence of apoptosis
- PMID: 11000227
- PMCID: PMC112387
- DOI: 10.1128/jvi.74.20.9562-9570.2000
Reovirus-induced G(2)/M cell cycle arrest requires sigma1s and occurs in the absence of apoptosis
Abstract
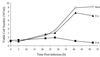
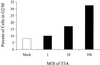
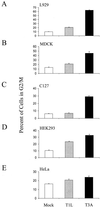
Serotype-specific differences in the capacity of reovirus strains to inhibit proliferation of murine L929 cells correlate with the capacity to induce apoptosis. The prototype serotype 3 reovirus strains Abney (T3A) and Dearing (T3D) inhibit cellular proliferation and induce apoptosis to a greater extent than the prototype serotype 1 reovirus strain Lang (T1L). We now show that reovirus-induced inhibition of cellular proliferation results from a G(2)/M cell cycle arrest. Using T1L x T3D reassortant viruses, we found that strain-specific differences in the capacity to induce G(2)/M arrest, like the differences in the capacity to induce apoptosis, are determined by the viral S1 gene. The S1 gene is bicistronic, encoding the viral attachment protein sigma1 and the nonstructural protein sigma1s. A sigma1s-deficient reovirus strain, T3C84-MA, fails to induce G(2)/M arrest, yet retains the capacity to induce apoptosis, indicating that sigma1s is required for reovirus-induced G(2)/M arrest. Expression of sigma1s in C127 cells increases the percentage of cells in the G(2)/M phase of the cell cycle, supporting a role for this protein in reovirus-induced G(2)/M arrest. Inhibition of reovirus-induced apoptosis failed to prevent virus-induced G(2)/M arrest, indicating that G(2)/M arrest is not the result of apoptosis related DNA damage and suggests that these two processes occur through distinct pathways.
Figures




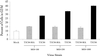

Similar articles
-
Linkage between reovirus-induced apoptosis and inhibition of cellular DNA synthesis: role of the S1 and M2 genes.J Virol. 1996 Nov;70(11):7984-91. doi: 10.1128/JVI.70.11.7984-7991.1996. J Virol. 1996. PMID: 8892922 Free PMC article.
-
Reovirus infection activates JNK and the JNK-dependent transcription factor c-Jun.J Virol. 2001 Dec;75(23):11275-83. doi: 10.1128/JVI.75.23.11275-11283.2001. J Virol. 2001. PMID: 11689607 Free PMC article.
-
Reovirus growth in cell culture does not require the full complement of viral proteins: identification of a sigma1s-null mutant.J Virol. 1998 Nov;72(11):8597-604. doi: 10.1128/JVI.72.11.8597-8604.1998. J Virol. 1998. PMID: 9765398 Free PMC article.
-
Mechanisms of reovirus bloodstream dissemination.Adv Virus Res. 2013;87:1-35. doi: 10.1016/B978-0-12-407698-3.00001-6. Adv Virus Res. 2013. PMID: 23809919 Free PMC article. Review.
-
Reoviruses and the host cell.Trends Microbiol. 2001 Nov;9(11):560-4. doi: 10.1016/s0966-842x(01)02103-5. Trends Microbiol. 2001. PMID: 11825717 Review.
Cited by
-
In Vitro Anti-cancer Activity of Adipose-Derived Mesenchymal Stem Cells Increased after Infection with Oncolytic Reovirus.Adv Pharm Bull. 2021 Feb;11(2):361-370. doi: 10.34172/apb.2021.034. Epub 2020 Apr 20. Adv Pharm Bull. 2021. PMID: 33880359 Free PMC article.
-
Reovirus nonstructural protein sigma1s is required for establishment of viremia and systemic dissemination.Proc Natl Acad Sci U S A. 2009 Nov 24;106(47):19986-91. doi: 10.1073/pnas.0907412106. Epub 2009 Nov 6. Proc Natl Acad Sci U S A. 2009. PMID: 19897716 Free PMC article.
-
Murine coronavirus replication induces cell cycle arrest in G0/G1 phase.J Virol. 2004 Jun;78(11):5658-69. doi: 10.1128/JVI.78.11.5658-5669.2004. J Virol. 2004. PMID: 15140963 Free PMC article.
-
Reovirus Activated Cell Death Pathways.Cells. 2022 May 27;11(11):1757. doi: 10.3390/cells11111757. Cells. 2022. PMID: 35681452 Free PMC article. Review.
-
The reovirus sigma1s protein is a determinant of hematogenous but not neural virus dissemination in mice.J Virol. 2011 Nov;85(22):11781-90. doi: 10.1128/JVI.02289-10. Epub 2011 Sep 14. J Virol. 2011. PMID: 21917967 Free PMC article.
References
-
- Advani S J, Brandimarti R, Weichselbaum R R, Roizman B. The disappearance of cyclins A and B and the increase in activity of the G(2)/M-phase cellular kinase cdc2 in herpes simplex virus 1-infected cells require expression of the alpha22/U(S)1.5 and U(L)13 viral genes. J Virol. 2000;74:8–15. - PMC - PubMed
-
- Braunagel S C, Parr R, Belyavskyi M, Summers M D. Autographa californica nucleopolyhedrovirus infection results in Sf9 cell cycle arrest at G2/M phase. Virology. 1998;244:195–211. - PubMed
-
- Brown E G, Nibert M L, Fields B N. The L2 gene of reovirus serotype 3 controls the capacity to interfere, accumulate deletions and establish persistent infection. In: Compans R W, Bishop D H L, editors. Double-stranded RNA viruses. New York, N.Y: Elsevier; 1983. pp. 275–287.
-
- Ceruzzi M, Shatkin A J. Expression of reovirus p14 in bacteria and identification in the cytoplasm of infected mouse L cells. Virology. 1986;153:35–45. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

